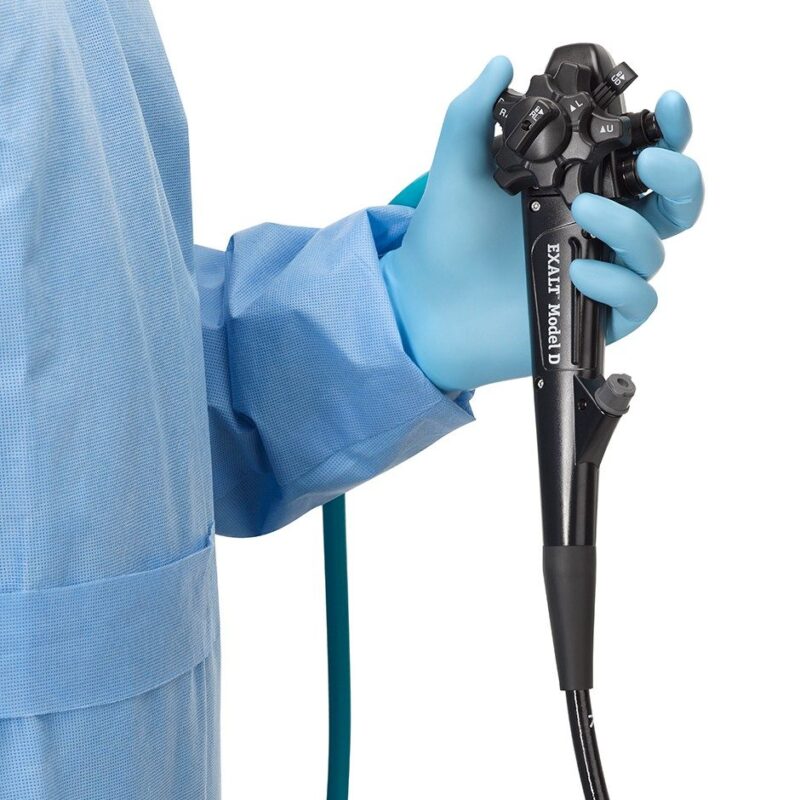
Boston Scientific (NYSE: BSX) announced that the U.S. Centers for Medicare & Medicaid Services (CMS) granted a New Technology Add-on Payment (NTAP) for single-use duodenoscopes, applicable to the EXALT™ Model D Single-Use Duodenoscope, as part of its Fiscal Year 2022 Hospital Inpatient Prospective Payment System.
NTAP was created to facilitate patient access for qualifying new medical technologies that substantially improve the diagnosis or treatment of Medicare beneficiaries. Beginning October 1, 2021, CMS will provide hospitals with additional device reimbursement when the EXALT Model D Single-Use Duodenoscope is used for eligible cases in the hospital inpatient setting.
The EXALT Model D Single-Use Duodenoscope is designed to eliminate the risk of infection due to ineffective reprocessing of traditional reusable duodenoscopes. Duodenoscopes are used to diagnose and treat various pancreatic and biliary conditions during endoscopic retrograde cholangiopancreatography (ERCP) procedures. Every year, more than 700,000 ERCP procedures are performed in the U.S. and approximately 1.5 million are completed worldwide.i In 2019 the U.S. Food and Drug Administration (FDA) recommended that providers utilize duodenoscopes with disposable components or fully disposable devices, when available.ii
The EXALT Model D Single-Use Duodenoscope previously received Breakthrough Device Designation from the FDA as well as transitional pass-through (TPT) payment by CMS, the latter of which allows for incremental device reimbursement for Medicare cases performed in the hospital outpatient setting. Further, in response to a Boston Scientific request, CMS created two unique ICD-10 procedure codes for hospitals to facilitate reporting the use of single-use duodenoscopes when used in the hospital inpatient setting beginning October 1, 2021.
“Securing NTAP, in addition to TPT, for the EXALT Model D Duodenoscope will support health care providers in accessing this device for Medicare patients – a group that represents over 40% of all ERCPs performed in the U.S. each year,” said Dave Pierce, executive vice president and president, MedSurg and president, Endoscopy, Boston Scientific. “Adequate reimbursement is key for the adoption of any new technology, and this decision from CMS will help provide hospitals with the means to treat Medicare patients with this device.”
The EXALT Model D Duodenoscope is part of the Boston Scientific single-use device portfolio, which includes technologies within the gastrointestinal, pancreaticobiliary, surgical, urological and airway spaces such as the LithoVue™ Digital Flexible Ureteroscope, SpyGlass™ DS Direct Visualization System, SpyGlass™ Discover Digital Catheter, and EXALT™ Model B Single-Use Bronchoscope.iii
For additional information, please visit here.