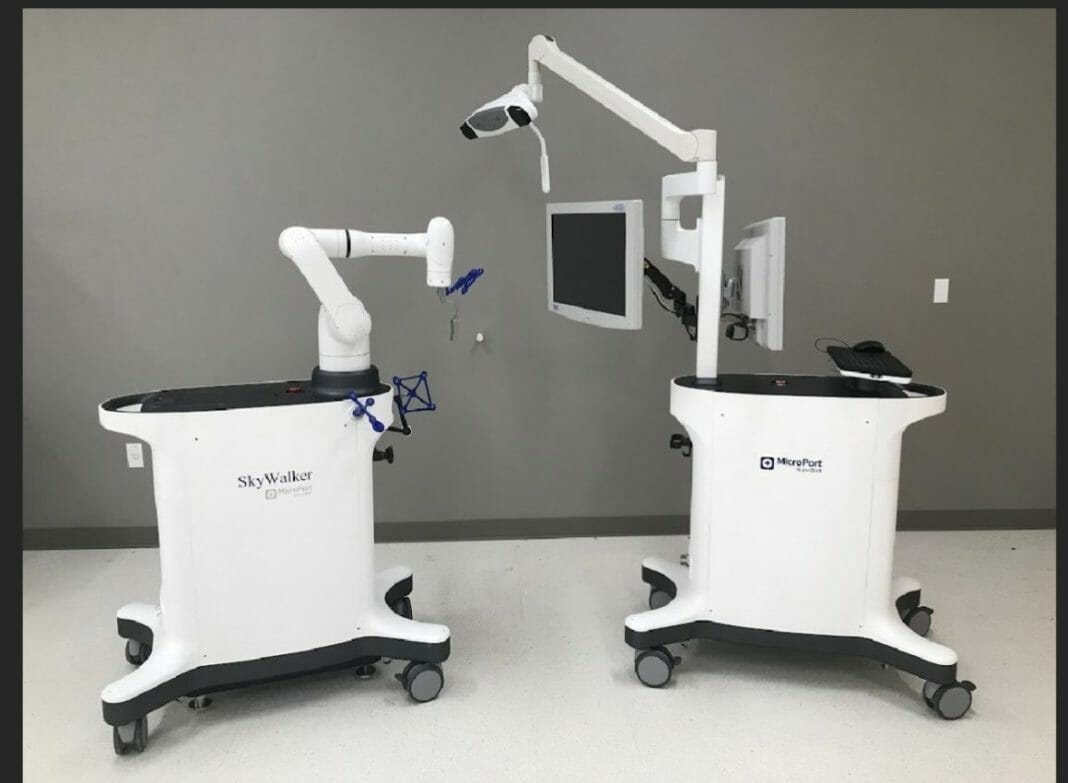
MicroPort Navibot has received 510(K) clearance from the Food and Drug Administration (FDA) in the United States for the SkyWalker™ System, the company’s first robot-assisted platform for orthopedic applications. SkyWalker will initially offer a robotically assisted total knee replacement solution that is compatible with the Evolution® Medial-Pivot total knee system.
Designed based on clinical needs, MicroPort NaviBot has built a full technology research facility that has medical industry design capability, and benefits from core technological advantages of having a self-developed, high-dexterity, and lightweight mechanical arm combined with intelligent planning and navigation algorithms. SkyWalker has the technical advantages of precise operation and efficient coordination, while prioritizing safety. Prior to the actual surgery, the planning system assists surgeons in formulating personalized patient implant plans based on preoperative CT scan anatomical data as well as specific implant data. During surgery, based on actual patient alignment and anatomy, the SkyWalker system facilitates precise implant positioning to achieve the desired kinematics specific to the patient. SkyWalker then allows the surgeon to quickly proceed to resection using a high-dexterity and lightweight mechanical arm resulting in improved precision, accuracy, and efficiency.
SkyWalker is capable of providing the surgeon with information that can help to achieve the desired joint line reconstruction while providing data to optimally balance soft tissues. MicroPort Navibot is planning to develop other orthopedic applications in the near future, in conjunction with MicroPort Orthopedics, for a more comprehensive orthopedic offering.