Next-generation cfDNA assay built on the Kihealth InterceptIQ™ platform expands the potential for earlier detection, dynamic disease monitoring, and precision therapeutic development in diabetes and metabolic disease
Medical Device News Magazine is committed to delivering high‑quality, in‑depth medical device news and biotechnology updates that include expert bylines. Our coverage offers a comprehensive view of the trends, innovations, and breakthroughs shaping the future of medical technology.
We also strive to provide a distinctive perspective on the rapidly evolving healthcare landscape. By staying connected with our reporting, you gain access to exclusive insights on industry developments, emerging technologies, and global regulatory changes that influence the medical device sector. At the same time, you join a vibrant community of professionals, innovators, and thought leaders dedicated to advancing healthcare through cutting‑edge solutions.
We invite you to explore our wide range of content—from the latest product announcements to detailed analyses of industry trends. And if our mission aligns with your goals, we offer opportunities for advertising and collaboration, helping you connect with a highly engaged audience of medical technology professionals.
Definition medical devices.
Medical Device Industry News | Latest Developments, Breakthroughs & Future Outlook
Latest News
E Tech Group Sees Rising Demand for IT/OT Assessments as Manufacturers Prepare Infrastructure for AI and Modernization
Manufacturers are prioritizing infrastructure visibility, cybersecurity, and operational readiness to advance modernization, AI initiatives and reduce hidden production risk
Ventris Medical Announces the Award of Additional Patent Claims for its Novel Backpack Bone Graft Containment System
Russell Cook, CEO of Ventris Medical - "This patent validates years of focused R&D and represents a meaningful step forward in solving one of the biggest challenges in bone grafting—maintaining the graft exactly where the surgeon places it
Salaera Named Halma Healthcare Company of the Year
The recognition honors the company's strong growth, strategic transformation and leadership in respiratory and gas technology
Clinical Trials
Paragonix Technologies Announces Launch of CONCERT-DCD Clinical Trial in Collaboration with UW Medicine to Maximize Heart Transplant Utilization
Prospective, multicenter trial will evaluate a novel preservation technique utilizing the Paragonix SherpaPak® for DCD (Donation after Circulatory Death) hearts
Combat Medical highlights patient impact of HIVEC HEAT trial at the American Urological Association Annual Meeting
The ongoing HIVEC HEAT trial aims to generate clinical data to evidence the efficacy of hyperthermic intravesical 80mg mitomycin C chemotherapy for patients with BCG-unresponsive, high risk NMIBC
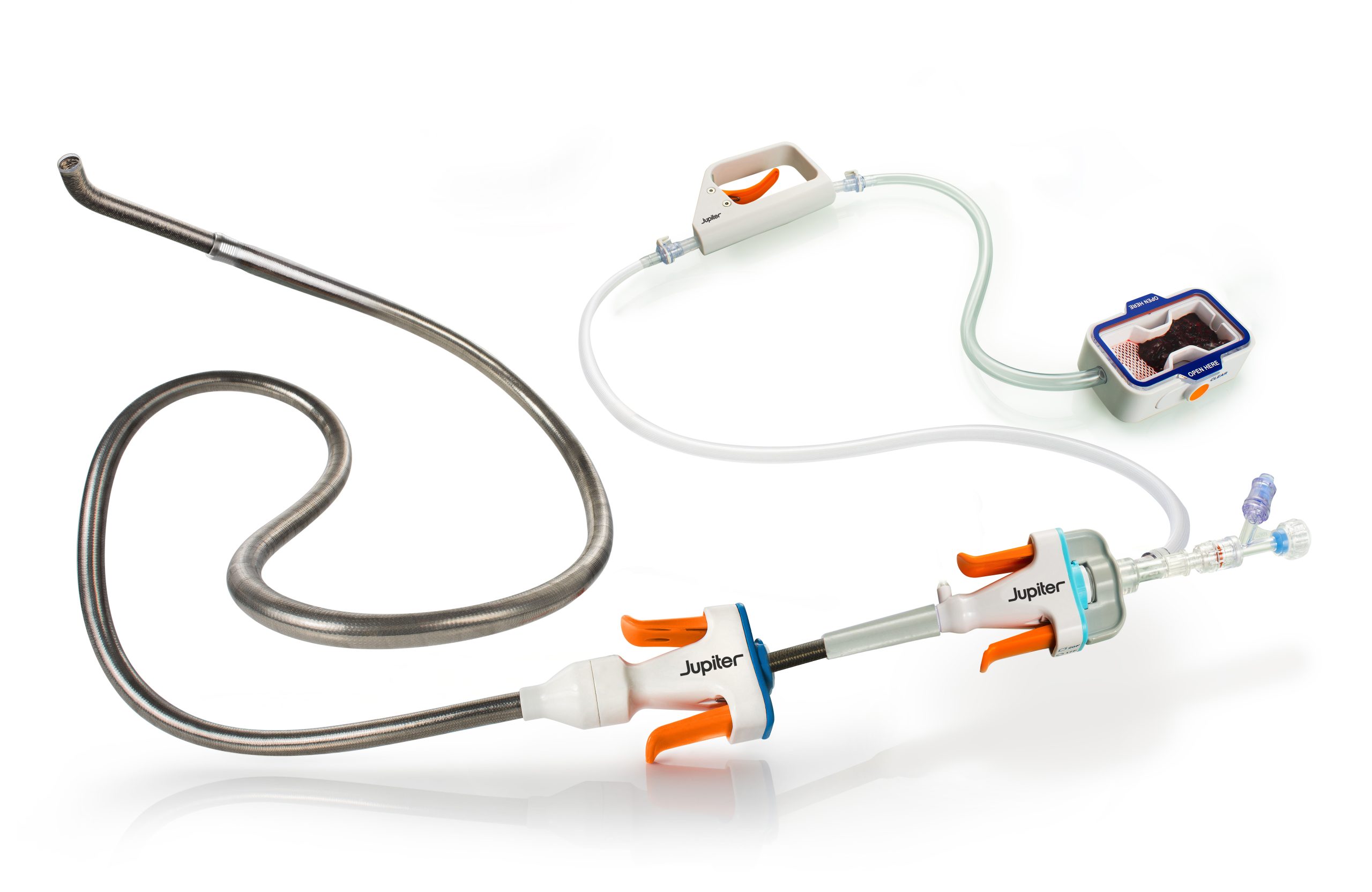
Jupiter Endovascular Reports Positive Results from SPIRARE II Pivotal Trial
Vertex Pulmonary Embolectomy System demonstrated significant right heart recovery, low adverse event rates, and reproducible performance across high-severity PE patients
Biotechnology News
JJP Biologics Announces Positive Interim Phase 1b Data for Nebaprubart (JJP-1212), an Anti-CD89 Antagonist, in Linear IgA Disease (LAD)
Favorable safety and tolerability profile, with rapid onset of clinical activity and therapeutic effects observed in patients with LAD
Wegovy® pill launches in the UAE as Novo Nordisk expands global access to obesity care
Novo Nordisk launches Wegovy® pill in the United Arab Emirates today
Lonza and Oxford Nanopore Technologies Launch Direct RNA Sequencing Solution for GMP mRNA Quality Control
“By integrating Oxford Nanopore’s direct RNA sequencing into Lonza’s QC environment, we are enabling a more comprehensive and scalable approach to mRNA quality control,” said Francis Van Parys, CEO, Oxford Nanopore Technologies
QIAGEN to advance AI-driven drug discovery with graph-based AI and curated bioinformatics knowledge with NVIDIA
Collaboration integrates NVIDIA accelerated computing and the NVIDIA BioNeMo platform with QIAGEN Digital Insights’ curated biomedical knowledge bases
Mergers & Acquisitions
Ernest Health Acquires Reunion Rehabilitation Hospitals, Expanding National Network
Upon completion of the transaction, Ernest Health will expand its footprint from 38 to 45 medical rehabilitation hospitals across the United States
Motivity Signs Definitive Agreement to Acquire Calmanac, Taking Full Ownership of the ABA Stack It Has Been Building
What started as a technical integration, connecting Motivity's gold-standard clinical data collection with Calmanac's enterprise-tested practice management capabilities, has since grown into something more: a shared understanding of what ABA practices are actually missing and a shared conviction about how to solve it.
Medtronic announces intent to acquire SPR Therapeutics, Inc., expanding care options for people living with chronic pain
Acquisition will enhance Medtronic's Neuromodulation portfolio with temporary peripheral nerve stimulation (PNS) technology, enabling earlier intervention for chronic pain sufferers
Funding
Ingenix Raises €13m From Sofinnova Partners-Led Syndicate to Scale Modality Fusion, a Novel Architecture for Drug Development
Ingenix's Biological Reasoning Engine fuses biological data through Modality Fusion, a new architecture for AI in drug development
Advanced NanoTherapies Secures Over $31M Series B to Deliver First-of-its-Kind Dual-Drug (Paclitaxel and Sirolimus) Nanoparticle-Coated Balloon Platform for Vascular Treatment
Financing supports the approval of U.S. Investigational Device Exemption (IDE), coronary clinical advancement, and expansion into below-the-knee peripheral (BTK) applications
Secretome Therapeutics Announces $30 Million Series A Financing
From RA Capital Management to Advance STM-01 for Duchenne-Associated Cardiomyopathy
Bylines by the Experts
By Yuri Nikolsky, Ph.D., D.A.B.R.M. University of Sharjah, Department of Medicine. University City, Sharjah, UAE. ynikolsky@sharjah.ac.ae
By: David Garratt, Principal Consultant - Automated IT Governance, Risk, Compliance, Verista
Katherine Klyushnichenko DiogeneAge, Inc. 1910 Thomes Avenue, Cheyenne, Wyoming 82001-3527, USA | kathy@diogeneage.com
While many regenerative treatments rely on injections administered in specialized clinics, his cream is designed to offer a non-invasive alternative
Cercare Medical Receives FDA 510(k) Clearance for Cone-Beam CT Perfusion
The clearance marks an important step forward in acute stroke imaging by bringing advanced perfusion analysis directly into the angio suite, allowing clinicians to assess brain tissue in real time during and immediately after thrombectomy procedures
TaeWoong Medical USA Receives FDA 510(k) Clearance for SPAXUS™, Expanding Its EUS Therapeutic Portfolio
The FDA determined the SPAXUS™ Stent to be substantially equivalent to legally marketed predicate devices, allowing for commercialization in the United States
Executives In the News
Lisa Nibauer Appointed CEO of NOW Diagnostics
Nibauer will lead next phase of innovation and transformation
ACU-Serve Names Natasha Baria Mehta President to Lead Next Phase of Strategic Growth
Mehta brings 25+ years of healthcare Revenue Cycle Management experience with a proven track record of scaling complex operations, driving financial performance, implementing advanced technologies, and building high-performing teams
Eko Health Appoints Dr. Steven Steinhubl as Chief Medical Officer
As Chief Medical Officer, Dr. Steinhubl will help drive Eko’s clinical strategy, research initiatives, and external scientific collaborations, further strengthening the evidence base and accelerating AI adoption across health systems worldwide.
Stealth BioTherapeutics Appoints David A. Brown, Ph.D., as Chief Scientific Officer
Brown brings more than 20 years of experience in mitochondrial research and scientific team leadership.
Advancements In Imaging
WellSpan Health and Philips Announce Landmark Strategic Alliance, Accelerating Innovation and Research Across Central Pennsylvania and Northern Maryland
Seven-year collaboration puts WellSpan at the forefront of diagnostics and imaging in community health through a groundbreaking research and innovation strategy aimed at shaping the future of care delivery
Philips receives FDA 510(k) clearance for Elevate Plus for EPIQ Elite and Affiniti, delivering AI advancements in general imaging ultrasound
AI-powered workflow automation helps clinicians acquire high-quality reproducible images faster and with greater confidence
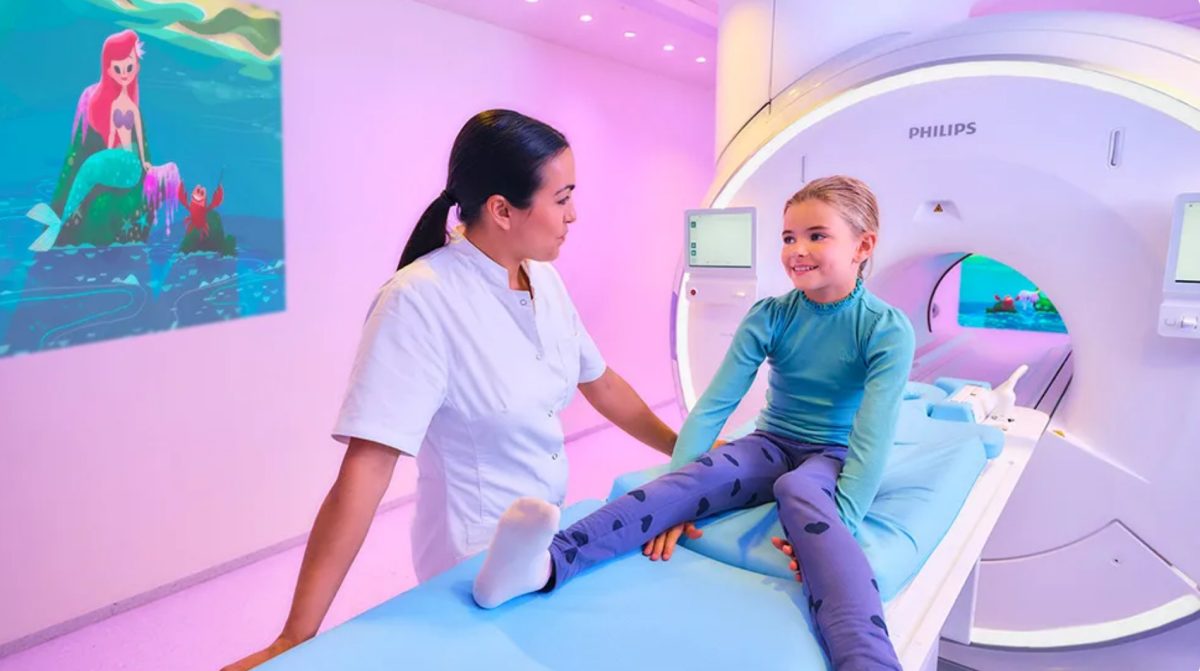
Disney and Philips bring together beloved storytelling and innovative technology to support kids undergoing MRI exams
Lisa Haines, Senior Vice President, Corporate Social Responsibility, The Walt Disney Company. “We’re proud to collaborate with Philips to extend that impact into MRI rooms in a meaningful way, using our beloved stories and characters to help provide moments of escape, normalcy, and reassurance during what can be an intimidating experience for kids in hospitals.”
Brainomix and Boehringer Ingelheim Advance Strategic Partnership in Pulmonary Fibrosis
Dr. Michalis Papadakis, CEO and Co-Founder of Brainomix said: “We are excited to expand our strategic partnership with Boehringer Ingelheim, a recognized leader and innovator in this field, with whom we share a firm commitment to improving outcomes for people living with pulmonary fibrosis.
Hospitals In the News

Delray Medical Center First Hospital in Palm Beach County to Use Spiration™ Valve System and SeleCT™ Screening for the Treatment of Severe Emphysema
SeleCT Screening automatically reviews existing chest computed tomography (CT) scans to help identify patients who would benefit from bronchoscopic lung volume reduction (BLVR). BLVR with endobronchial valves such as the Spiration Valve System may improve lung function by allowing airflow to be redirected from hyperinflated portions of the lung to healthier portions
Chapters Health System Announces Selection on CHROs to Know List
Chief People Officer, Nikki Romence, has been named one of Becker’s Hospital Review’s “140 Hospital and Health System CHROs and Chief People Officers to Know” for 2026
Baptist Health, Compass Surgical Partners Advance Northeast Florida Surgery Network with Middleburg Surgery Center
Located in southeast Jacksonville near Baptist Medical Center Clay, the multispecialty ASC supports procedures in orthopedics, urology, otolaryngology, pain management, and general surgery. With 15 physician partners committed to the center, the partnership is evaluating expansion opportunities to meet growing demand.
First Multi Functional European Training in Augmented SBRT conducted in Monza
The clinic is currently conducting research into single-fraction treatment, where therapy is delivered in one session (while traditional radiotherapy requires 20–40 treatment sessions)
Non-Profit Association News
NMDP Celebrates Public-Private Partnership with HRSA in Expanding Patient Access to Life-Saving Bone Marrow and Cord Blood Transplant
Nonprofit reaches milestone of facilitating 150,000 transplants giving patients a second chance at life, thanks to federal investment in and Congressional commitment to the C.W. Bill Young Cell Transplantation Program
AHF Reports: Ebola Crisis Exposes Failure of Current Global Health Architecture
The current outbreak involves the Bundibugyo strain of Ebola virus, for which no approved vaccines or therapeutics currently exist, heightening concerns among global health officials and frontline responders, according to reporting by Health Policy Watch
Health Leaders Join Forces to Launch First-of-Its-Kind Women’s Health AI Consortium
Industry leaders from across the health ecosystem, including Clue, Ema EQ, Thrive Global, Oura, and Willow, will set the first clinically rigorous, transparent standards for AI in women’s health
Healthcare: Understanding the Business Site of the Healthcare Industry and Its Implications
The Digital Pulse: Synchronizing Healthcare Infrastructure, Advanced Capital Modeling, and Technical Security
The structural architecture defining healthcare management is undergoing a fundamental, system-wide overhaul
The Risk of Misinformation in Health Content Marketing
Introduction Although there has always been health misinformation, its current scope, velocity, and impact are unmatched in contemporary times. Health misinformation is one of the biggest dangers to public health, according to a consensus statement published by the American Psychological Association in 2025. The World Health Organization has referred to this as...
How Digital Access Is Reshaping the Way Patients Choose Specialized Care
As technology continues to evolve, this trend will likely become even more pronounced. Patients will have access to more detailed information, better tools for comparison, and clearer insights into the care they’re considering.
Careers In Healthcare
HomeCEU Introduces New OT Grad Toolkit to Support Occupational Therapy Graduates Looking for Jobs in the Healthcare Industry
This employment toolkit is designed to walk a new occupational therapy (OT) graduate through the process of landing their first job in healthcare
How to Evaluate the Quality of a Graduate Business Program
Choosing a graduate business program today is not just about prestige. It is about outcomes, flexibility, and relevance in a world shaped by AI, remote work, and economic uncertainty.
How Education Can Open Doors in Healthcare Professions
Education is more than a requirement in healthcare; it is the key that unlocks opportunity, stability, and purpose.
Nurse's Corner
Accelerated BSN Programs: A Swift Solution to the Nursing Shortage
Accelerated BSN programs offer a rapid pathway into nursing for those with prior degrees and these programs help address the nationwide nursing shortage by quickly preparing qualified professionals.
Nurse Burnout: A Strategic Threat Hospitals Can’t Afford to Ignore
Burnout isn’t just “being tired.” It’s a chronic state of emotional, mental, and physical exhaustion caused by prolonged stress.
Nurses | How To In Shape
Many nurses manage to stay in great shape, both physically and mentally—despite the challenges