Medline Announces FDA Clearance for REFLEX HYBRID Nitinol Implants for Foot and Ankle Surgery
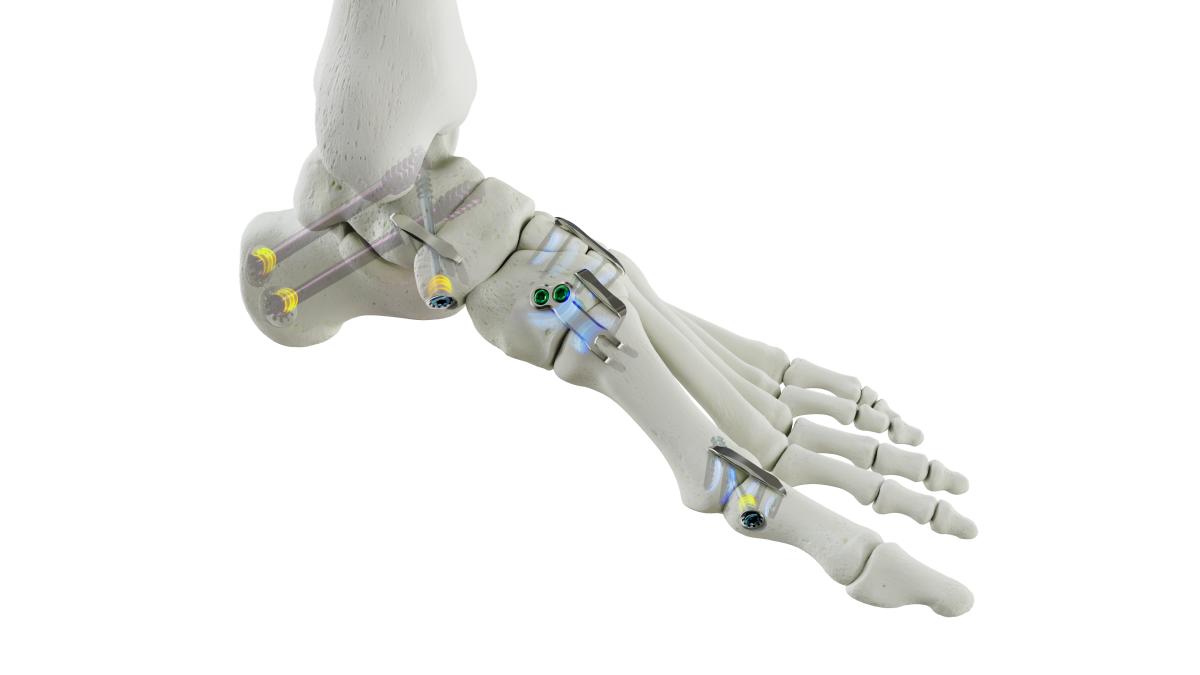
“REFLEX HYBRID further demonstrates our commitment to offering innovative solutions for foot and ankle surgeons. The first to market product addresses gaps in the current competitive landscape, including offering indication specific designs, intraoperative compression, and intraoperative adjustment with a nitinol implant,” said Scott Goldstein, vice president of product management for Medline UNITE.