Medtronic receives FDA clearance for Stealth AXiS™ surgical system | 1st Integrated planning, navigation and robotics platform for spine surgery
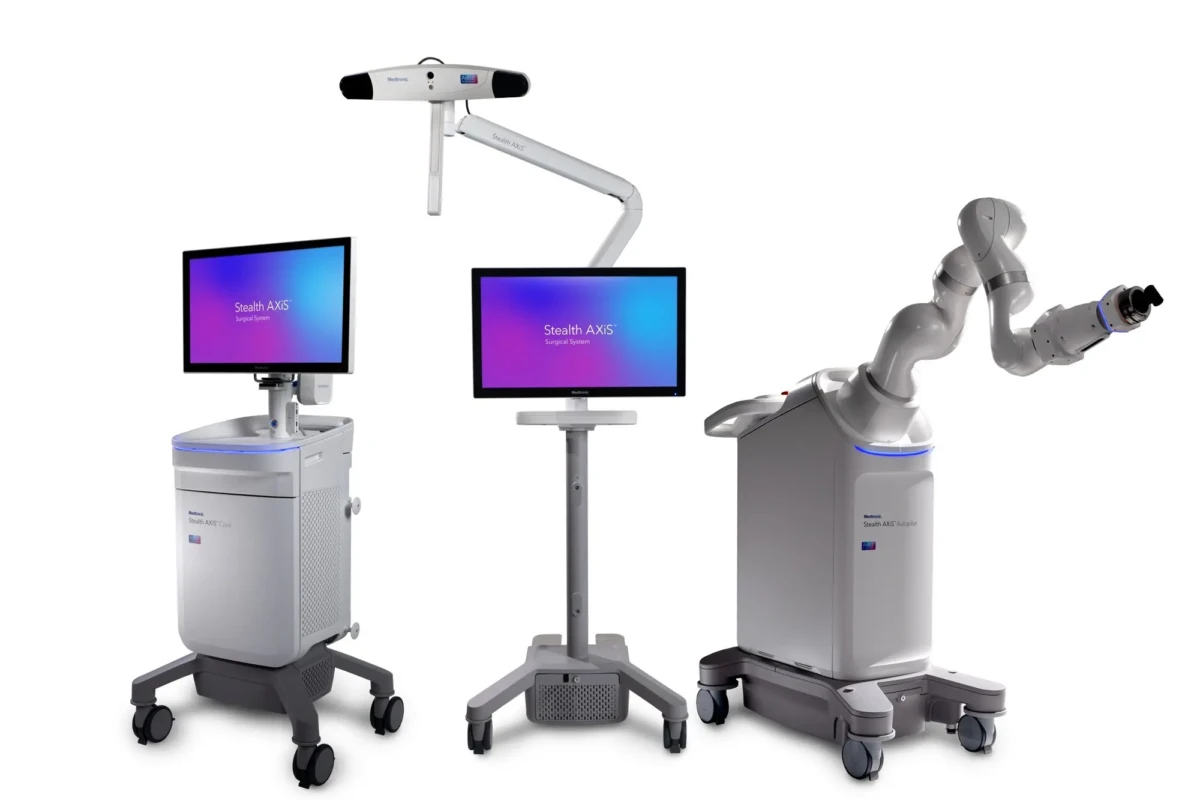
The Stealth AXiS™ system is cleared for spine procedures in the United States, with an underlying architecture designed to support future cranial and ENT applications, pending 510(k) clearance.