
Hypertension, defined as having a blood pressure at or above 130/80 mm Hg, has often been called the silent killer, putting a person at increased risk of heart disease, heart failure, stroke, and many other conditions.
In 2023 alone, high blood pressure was a primary or contributing cause of 664,470 deaths in the United States, according to the U.S. Centers for Disease Control (CDC).Nearly half of adults in the U.S. have high blood pressure (119.9 million people) and 3 in 4 do not have it under control.
Despite it being the costliest disease and the most prevalent chronic condition in the country, it remains underdiagnosed, undertreated, and frequently misunderstood. The primary reason is measurement strategy.
How patients and providers can stem the tide
An episodic in-office test is clearly not sufficient for tracking the onset and progression of hypertension which is increasingly becoming an epidemic.
Conducting a once-a-year blood pressure check will certainly miss what happens in between.
The variability of blood pressure numbers require consistent monitoring and tracking to provide any meaningful diagnostic insight for physicians and patients.
Providers and researchers have focused on this problem for years and a multitude of innovations have sought to address this issue, however challenges have persisted.
The cuff solution, while having been the gold standard for years, is impractical, especially when one considers that round-the-clock ambulatory blood pressure monitoring is really what is needed to effectively diagnose conditions such as “white coat” syndrome and masked hypertension.
While blood pressure monitoring solutions in health clinics have evolved from manual (mercury-based devices) and are way ahead of where they used to be, current options are still sub-optimal, bringing with them effects like sleep disruption, ultimately compromising quality of life and resulting in poor patience adherence.
Effective blood pressure control, particularly monitoring blood pressure, is critical to preventing or reversing structural changes caused by high blood pressure.
Using AI and Machine Learning to Accurately Assess Cardiovascular Risk
Left ventricular hypertrophy (LVH), clinically defined by an elevated left ventricular mass index (LVMI), is a strong predictor of adverse cardiac outcomes. Currently, measurements of LVMI are performed using transthoracic echocardiography, accessible only in a clinical setting.
A scientific research paper by authors from Columbia University, published by Nature’s NPJ Biosensing, used data from an ambulatory blood pressure monitor, combined with laboratory test results and demographics data collected one time, initially, to investigate whether LVMI and LVH may be estimated and classified accurately.
In the study, researchers developed and tested machine learning models using clinical data (including data from the Framingham Heart Study (FHS) and waveform data from the Masked Hypertension Study (MHTN).
The study presented a method for accurately assessing cardiovascular risk using ambulatory measurements and highlighted the use of waveform data for assessing hypertrophy.
Considering research showing that individuals with masked hypertension have an increased risk of LVH, the ability to diagnose LVH, confirmed with echocardiography in masked hypertensives, would enable healthcare providers to prescribe hypertrophy management earlier than they would otherwise if caught at a clinic.
The Columbia University study’s classification models were able to discriminate LVH cases with an Area Under the Curve (AUC) of 0.95 or higher (and an AUC of 0.93 or higher for concentric LVH). By comparison, previously published LVH detection models used 12-lead ECGs for their analyses, with AUCs of 0.8240, 0.8726, 0.8941, and 0.9642. (In machine learning, AUC represents the degree or measure of separability. The higher the AUC, the better the model is at distinguishing between patients with the disease and no disease).
Waveform-derived parameters, such as reflection coefficients, provide additional information about left ventricular hypertrophy risk. For example, diastolic dysfunction is an early feature of hypertensive LVH1, and the blood pressure waveforms of patients with hypertensive LVH have been shown to phenotypically differ, due to wave reflections, from healthy waveforms.
Researchers concluded that the results of the study warranted further investigation into using blood pressure waveforms for cardiac risk assessment. It also indicated that deep learning can be used as a tool to enable early detection of cardiac risk using data from wearable devices.
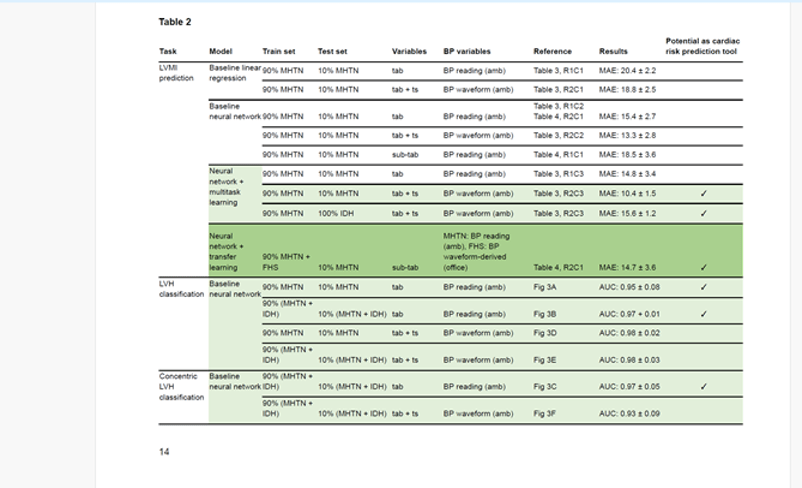
Table 2. Summary of all the tested models. Rows highlighted in light green meet the criteria for accurate approximations using ambulatory data, and the row in dark green is the transfer learning case which uses fewer variables for predictions. Rows with ‘Potential as cardiac risk prediction tool’ checkmark warrant further investigation towards being potentially suitable for ambulatory cardiac risk assessment. ‘amb’ is ambulatory data, ‘clin’ is clinic measurement. ‘R’ is the row number, ‘C’ is the column number. Cases with ‘tab’ under the ‘Variables’ column use all 37 variables (see Supplementary Data for list). Cases with ‘sub-tab’ use 7 variables (as seen in Supplementary Table 2). Cases with ‘tab + ts’ use all 37 tabular variables and waveforms (time series). MHTN is Masked Hypertension Study, FHS is Framingham Heart Study, IDH is Improving the Detection of Hypertension. Results are presented as mean ± standard deviation, MAE is mean absolute error, AUC is area under the curve.
From Behnam, Vira, Chern, Terry L., Wiedenhaupt, Jannik, Shimbo, Daichi, Schwartz, Joseph E., Sia, Samuel K, Assessment of cardiovascular risk from ambulatory blood pressure measurements enabled by deep learning npj Biosensing 2, 18, (2025) https://doi.org/10.1038/s44328-025-00037-y
But what about wrist-based wearable devices like the Apple Watch and other similar products?
Unfortunately, these devices, while offering the ability to track trends, aren’t there yet when it comes to providing the accuracy and consistency of data that physicians and healthcare providers require. From a physiological standpoint, the wrist is also an impractical location for reliable blood pressure assessment due to distance from the brachial artery, sensitivity to positioning relative to heart level, and susceptibility to motion artifacts. They can’t provide actual blood pressure numbers much less round-the-clock, diurnal and nocturnal data. In addition to the question of accuracy, there are also challenges related to calibration, arm positioning, and user variability.
According to the American Medical Association (AMA), factors such as body habitus, vascular stiffness and arrhythmias can affect measurement reliability, and some blood pressure monitoring devices perform better in younger, healthier populations than in elderly or critically ill patients.
The Impact of Cuffless Upper-Arm Ambulatory and Remote BP Monitoring Technology
Annual costs associated with high blood pressure were an estimated $219 billion in the United States in 2019 alone.
While Hypertension is a staggering epidemic, and highly costly, it is more preventable than many other conditions.
Technology can help by providing physicians and patients with the data they need to create better prevention and treatment plans and outcomes.
However, not every device is created equal.
Smart watches and fitness trackers are not adequate because they gather other biological data from a person’s body and use that to approximate their blood pressure. These devices are useful to provide directional screening, but they have limitations that leave a lot to be desired. They can only measure relative changes in blood pressure, not absolute numbers.
Also, these types of devices don’t have methodologies to make corrections for movement, making it useless for physicians.
As explained in a recent Consumer Reports article, expert-developed protocols in regards to making medical decisions based on blood pressure measurements are for the most part built based on blood pressure readings taken at rest and under specific circumstances (one is sitting in the correct position, not talking, and after being at rest for a full 5 minutes beforehand, etc., for example).
Technologies are now under development, using AI and deep learning, for use in the clinical setting that can address a full spectrum of challenges that can come with remote blood pressure monitoring offered on rings, watches, and other consumer devices and even on cuff-based devices.
Still, this kind of innovation is not just transforming remote blood pressure monitoring. We are increasingly seeing a number of innovations that are leveraging AI and deep learning for remote patient monitoring and drug delivery in other indications as well.
For example, Abbot’s new Libre Assist, uses generative AI to predict how a food choice may affect a person’s glucose levels and then offers personalized meal guidance before a meal and leverages data from Abbot’s leading Libre CGM systems sensors to confirm the actual impact afterwards. Meanwhile U-Pump, which completed its MVP in April of last year, is developing its Insulin pump system with a small, lightweight design that brings advanced AI technology to monitor glucose levels and automatically adjust insulin delivery in real-time.
How to Monitor Real Blood Pressure Remotely Yet Unobtrusively
Technologies that leverage an upper-arm configuration – a band worn on the upper-arm – and one that incorporates photoplethysmography (PPG) sensors to measure pulse wave velocity and combine that with environmental sensors (barometer, accelerometer, etc.) and algorithms to estimate true blood pressure, without a cuff, are offering new options for patients and providers.
Upper-arm devices incorporating this technology use multiple components including a pulsewave detector, external‐pressure compensator, a blood pressure‐tracking and a calibration algorithm, and a stability detector to continuously adjust its readings while offering capabilities to adjust for comfort, ensure continuous tracking, and ensure accuracy regardless of arm position or movement.
Upper-arm devices have been found to be much more accurate than any devices worn on the wrist – whether it be a smartwatch or any other wrist-based wearable device cuff-based or otherwise. A wrist blood pressure monitor takes its measurements at the radial artery near where a person’s wrist meets their hand. An arm blood pressure monitor takes its measurements higher up, at a person’s brachial artery in their upper arm, which is the anatomical site most physicians are trained to use and are clinically familiar with for standardized blood pressure assessment.
While ambulatory blood pressure waveform monitors have traditionally not been as widely available as regular blood pressure cuffs that provide discrete readings, cuffless blood pressure monitoring devices that are configured to offer continuous, remote and ambulatory monitoring capabilities, day and night, are now increasingly becoming a viable option for overcoming challenges related to monitoring blood pressure in outpatient settings.
The clinical community will soon be able to take advantage of these and other novel innovations that make use of increasingly sophisticated AI models, waveform data, and analytics, furthering precision medicine to deliver better options and outcomes for patients at every hypertensive stage.
Then we will realize a future where Hypertensiveness no longer lurks in silence.
Editor’s Note: Dinesh Kumar, Ph.D. is Founder and CEO of Blend Health Technologies Inc. He leads the company in transforming hypertension management and bringing the world proactive healthcare with its cuffless, continuous blood pressure monitoring device. He is a C-suite executive, entrepreneur and startup advisor with a demonstrated history of commercializing novel medical technologies. Previously, he served in several foundational and executive leadership roles at life sciences and medical device companies including Neuro24 and Promaxo, among others. He earned an M.B.A. from the University of California, Berkeley, Haas School of Business and a Ph.D. in Electrical and Computer Engineering from The University of Iowa.


