As regulatory bodies like the FDA continue to update drug labeling with biomarkers, the role of high-precision medical devices will only grow
Medical Device News Magazine is committed to delivering high‑quality, in‑depth medical device news and biotechnology updates that include expert bylines. Our coverage offers a comprehensive view of the trends, innovations, and breakthroughs shaping the future of medical technology.
We also strive to provide a distinctive perspective on the rapidly evolving healthcare landscape. By staying connected with our reporting, you gain access to exclusive insights on industry developments, emerging technologies, and global regulatory changes that influence the medical device sector. At the same time, you join a vibrant community of professionals, innovators, and thought leaders dedicated to advancing healthcare through cutting‑edge solutions.
We invite you to explore our wide range of content—from the latest product announcements to detailed analyses of industry trends. And if our mission aligns with your goals, we offer opportunities for advertising and collaboration, helping you connect with a highly engaged audience of medical technology professionals.
Definition medical devices.
Medical Device Industry News | Latest Developments, Breakthroughs & Future Outlook
Latest News
BD Delivers Next‑Generation TIPS Innovation to Advance Portal Hypertension Care Across the European Union
CE Marking and ARCH clinical trial data position the Liverty™ TIPS Stent Graft to expand treatment options for patients with advanced liver disease
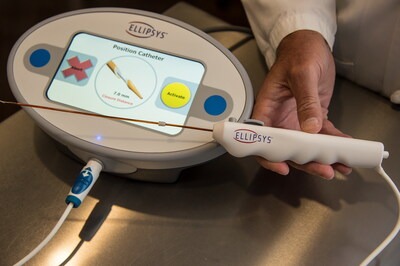
Ellipsys Vascular Access System to Return to U.S. Market in 2026
Ellipsys Medical, Inc. today announced that the Ellipsys® Vascular Access System will once again be commercially available, with initial availability anticipated in mid-2026. The announcement marks the planned reintroduction of the minimally invasive technology to clinicians and patients following a period of limited market availability. The Ellipsys System, previously available...
Clinical Trials
Phantom Neuro Receives Approval for First-In-Human Clinical Study
The study's PI is Michael Lo, M.D., a leading plastic surgeon at Cabrini Health
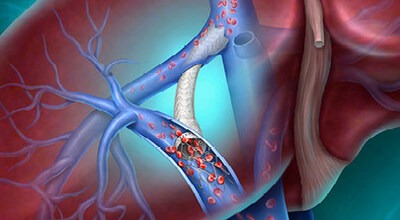
New STORM-PE Data Show Computer Assisted Vacuum Thrombectomy with Anticoagulation Resulted in Significantly Better Functional Outcomes at 90 Days Compared to Anticoagulation Alone for Pulmonary Embolism Patients
Mechanical Thrombectomy Dramatically Outperforms Standard PE Care at 90 Days
First Implant of KingstronBio’s ProStyle M® Transcatheter Mitral Valve System Successfully Completed in National Multicenter Confirmatory Study
On March 12, 2026, the first implant of the ProStyle M® Transcatheter Mitral Valve System (ProStyle M®) was successfully completed under pure ultrasound guidance as part of its confirmatory clinical study. The device is independently developed by KingstronBio Technology (Changshu) Co., Ltd. (KingstronBio). The procedure was performed by Professor Wang Chunsheng and Professor...
Biotechnology News
Tsingke Unveils ‘Zero-Contact’ Gene Synthesis to Safeguard Core Genetic Sequences
As the pace of biotechnology research accelerates, companies are increasingly challenged to balance rapid innovation with the protection of sensitive genetic information.
Variational AI Releases Enki 4: Major Update to Foundation Model for Small-Molecule Drug Discovery
Variational AI introduces Enki™ 4: an improved algorithm and architecture designed to scale, expanding the pre-trained target coverage from 592 to 760 and now applicable to proximity-based therapeutics and novel payloads for antibody drug conjugates
Transgene and NEC Bio Sign License Agreement to Prepare the Next Steps of the Development of TG4050 in Head and Neck Cancer
Akira Kitamura, GM, AI Drug Development Division of NEC Corporation and CEO of NEC Bio, added, "This agreement is an important milestone in our partnership with Transgene and reflects NEC’s long-term commitment to the development of TG4050
Mergers & Acquisitions
Funding
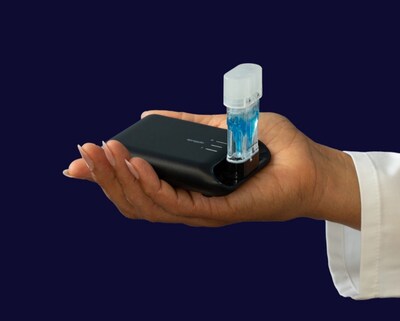
Aptitude Awarded a BARDA Contract Modification to Add Avian Flu Detection to the Metrix® Platform
This funding will support the development of Avian H5 Influenza detection capabilities on the Metrix® platform, expanding upon its recent U.S. Food and Drug Administration (FDA) emergency use authorized COVID-19/Flu multiplex test.
TA Invests in HealthMark Group to Support the Next Phase of Growth
As part of the transaction, HealthMark management and existing investor Ridgemont Equity Partners will both roll meaningful ownership stakes, welcoming TA as the new lead investor. Financial terms of the transaction were not disclosed.
Industry Expert Bylines

How AI Can Boldly Scale Cybersecurity Beyond Human Limits | By John Spencer-Taylor
Organizations must adopt a proactive defense strategy to stay ahead, which isn’t always easy to do with human capabilities that are limited. Fortunately, artificial intelligence can act as an auxiliary defense force and play a key role in helping improve cybersecurity efforts in a number of ways, responding faster than human teams alone.

A Wave of Drug-Free Alzheimer’s Treatment Innovation Offers New Hope | By Chuck Papageorgiou, CEO, NeuroEM Therapeutics
Chuck Papageorgiou writes: “With no effective prevention or long-term treatment, receiving a diagnosis of Alzheimer’s disease can seem like a fate worse than death for patients and their families and caregivers. However, a wave of innovation is bringing new hope to the 7 million Americans already living with Alzheimer’s disease and the 500,000 who will receive this devastating diagnosis each year.”

How AI is Transforming Vision Care | By Matthias Hofmann, Co-founder & CEO of Eyebot
“The way we think about healthcare is evolving rapidly. From AI-assisted diagnostics to telehealth expansion, technology is breaking down barriers and reshaping how people access essential medical services. In eyecare, artificial intelligence is increasingly redefining accessibility, affordability, and accuracy in vision care,” writes Matthias Hofmann.
FDA Medical Device Updates
Market Reports
FDA Grants HemoSonics 510(k) Clearance to Expand Use of Its Quantra Hemostasis System to Obstetric Procedures
he Quantra Hemostasis System provides comprehensive blood analysis that delivers rapid, real-time coagulation insights at the point of need, empowering clinicians with actionable data to make better decisions and protect mothers who experience postpartum hemorrhage during childbirth. Quantra is the first and only cartridge-based FDA-cleared viscoelastic testing platform for obstetrics
Medtronic secures FDA approval for the Altaviva™ device, a simple experience for treating urge urinary incontinence
“The Altaviva device represents a significant advancement for patients with urge urinary incontinence who often weigh the invasiveness of a therapy and the potential side effects when considering treatment options,” said Dr. Kevin Benson, Urogynecologist at Sanford Health and investigator for the Altaviva device pivotal study (TITAN 2)
Executives

Channel Medsystems Strengthens Commercial Leadership Team with Appointments of Tara Murphy as Vice President of Marketing and Paul Russo as Vice President of Sales
“We’re thrilled to welcome Tara and Paul to the Channel leadership team,” said Nelson Whistance, President and Chief Executive Officer of Channel Medsystems
Nanopath Expands Leadership Team to Drive Next Phase of Scientific and Commercial Growth
“Not only are these individuals expert in their respective fields, they are also highly collaborative, fantastic leaders, and embody the mission and culture of the company” said Alison Burklund, Ph.D., Co-Founder and Co-CEO at Nanopath
Italfarmaco Appoints Francesco Di Marco as CEO
Dr. Di Marco brings over 25 years of experience in the biopharmaceutical industry through multiple executive leadership positions
Advancements In Imaging

Philips and Mass General Brigham: Advancing Patient Care
Discover how Philips and Mass General Brigham are transforming healthcare with innovative AI solutions for better patient outcomes.

KA Imaging’s Premium Dual-Energy Mobile System Now Available For Sale In Europe Reveal Mobi Pro Has Received the CE Mark
The Reveal Mobi Pro ™ integrates KA Imaging’s Reveal™ 35C detector with SpectralDR® technology into a complete mobile X-ray solution.
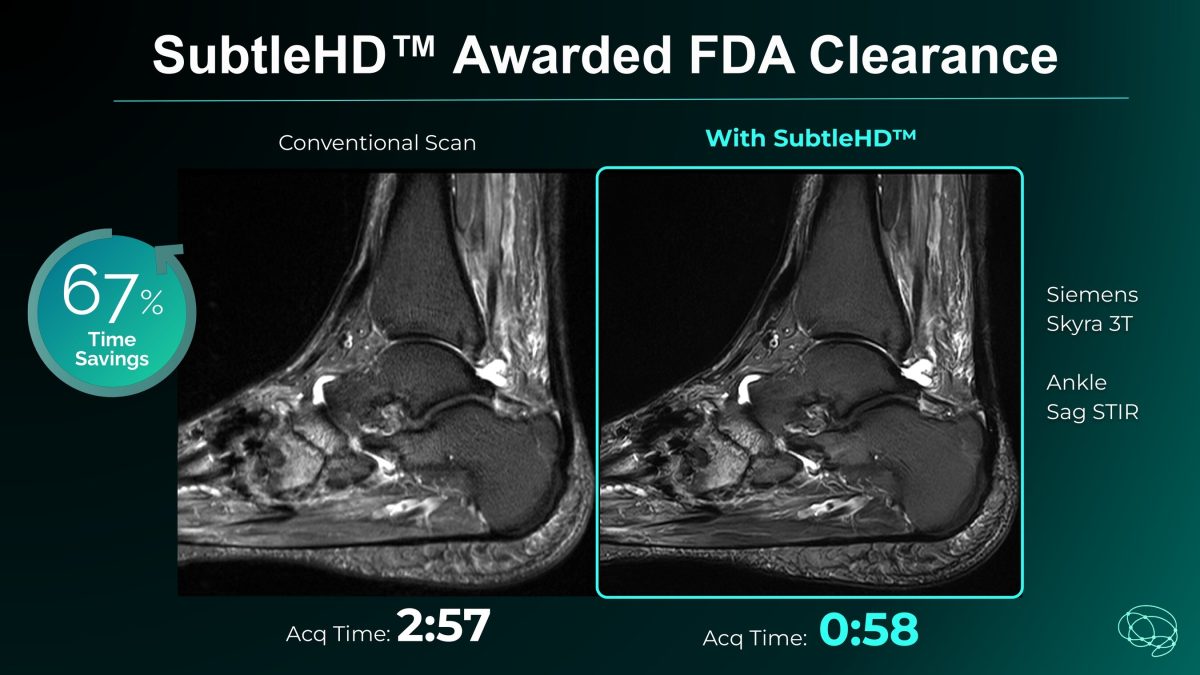
Subtle Medical Reports: SubtleHD™ Awarded FDA Clearance, Setting a New Benchmark for MRI Image Quality and Speed
With FDA clearance of SubtleHD™, Subtle Medical launches Subtle-ELITE™, an industry-first AI package designed to achieve superior image quality, unprecedented speed, and workflow automation.
Hospitals In the News

Marcus Neuroscience Institute Performs First Next-Generation Augmented Reality Spinal Surgery at Bethesda Hospital West
Marcus Neuroscience Institute, part of Baptist Health, at Bethesda Hospital West, is the first hospital in the state of Florida to perform and offer next-generation augmented reality spinal surgery with novel, advanced, high-resolution AR technology. The surgery was done by neurosurgeon Timothy O’Connor, M.D., director of minimally invasive and robotic spine surgery. Marcus Neuroscience Institute is one of the first medical centers in the world to use this next-generation augmented reality technology.
Drug Prevents Heart Failure After Heart Attacks in Mice, Study Finds
This discovery could lead to new treatments to prevent heart failure, a serious cardiac condition that develops in up to 30% of heart attack survivors within one year.
Smart Hospitals: Redefining Global Healthcare with Digital Innovation
Smart hospitals are at the forefront of a global transformation in healthcare, integrating advanced technologies to improve patient outcomes, enhance operational efficiency, and reduce costs.
Non-Profit News
Healthcare: Understanding the Business Side of the Industry and Its Implications

Why is AI Healthcare Scribe Essential for Clinicians in 2025
AI medical scribes play a crucial role for clinicians and patients. Traditional scribes create documentation and burnout crises, security and privacy issues, and poor communication between physicians and patients, while AI ambient scribes can effectively address documentation crises, reduce burnout, enhance patient experience and well-being, and offer cost-effective and scalable solutions

How to Effectively Use Digital Billboards
Advertising has come a long way since paper-painted signs were posted on the side of the road. Now, digital billboards convey your brand narrative more effectively than

Bleach-Infiniter in Chicago: Helping Clinics Compete Without Extra Costs
Learn how Bleach-Infiniter helps Chicago clinics enhance services without added expenses, boosting competitiveness in healthcare
How Staffing Platforms Improve Compliance in Healthcare
Maintaining compliance is one of the most important responsibilities healthcare facilities face—and one of the most complex