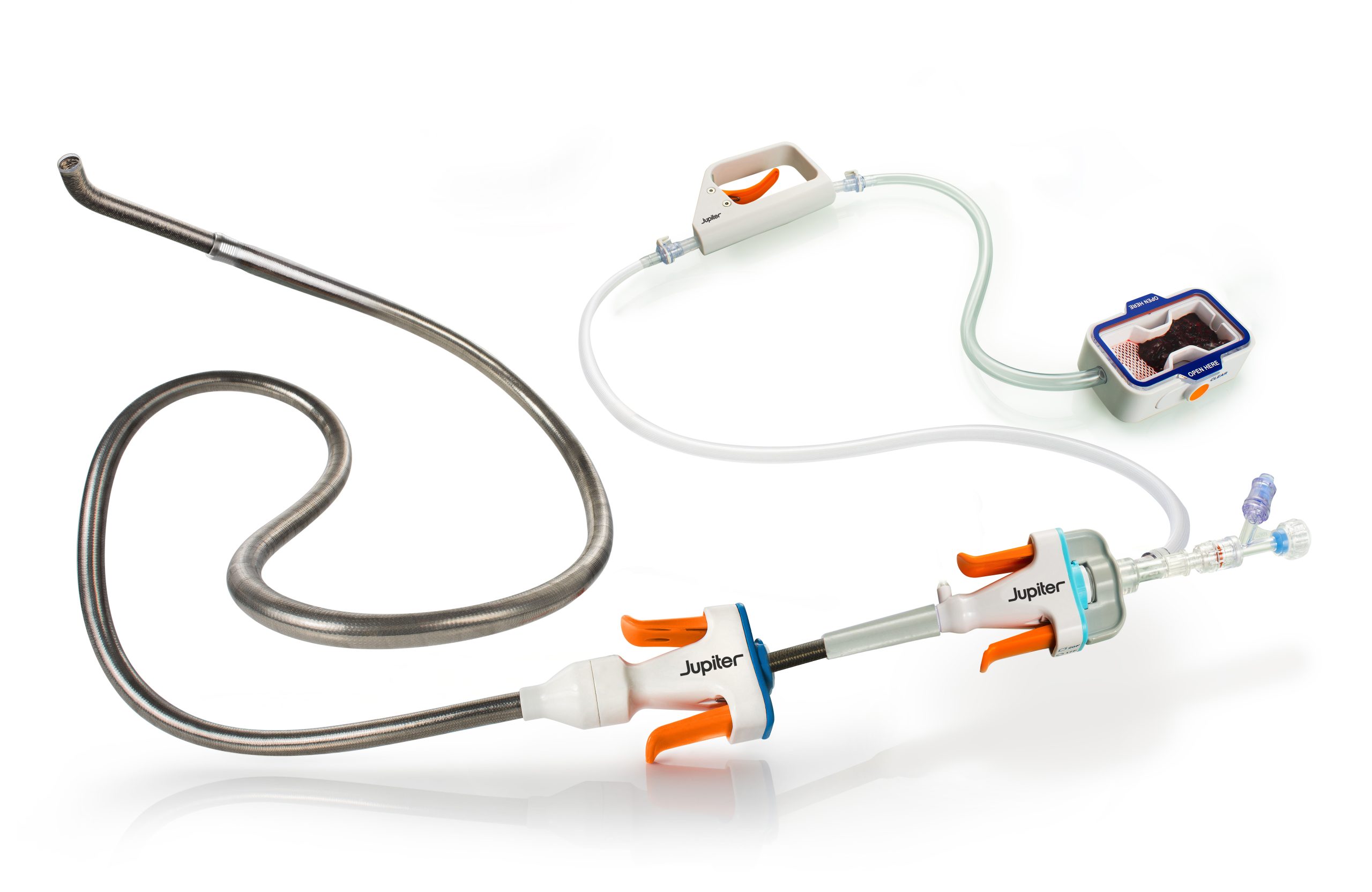
Vertex Pulmonary Embolectomy System demonstrated significant right heart recovery, low adverse event rates, and reproducible performance across high-severity PE patients
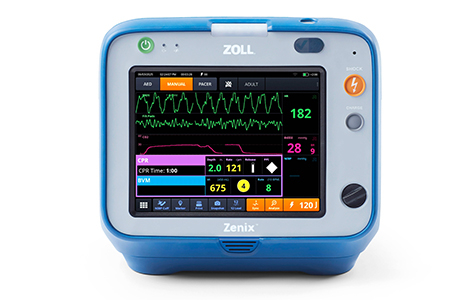
Asahi Kasei Elevates Critical Care Solutions with FDA Approval of ZOLL’s Zenix Monitor/Defibrillator
The Zenix monitor/defibrillator is designed for use in EMS and hospital settings








![IGT IVUS[18] - Medical Device News Magazine - Medical Device News Magazine IVUS](https://infomeddnews.com/wp-content/uploads/2024/01/IGT-IVUS18-400x267.jpg)