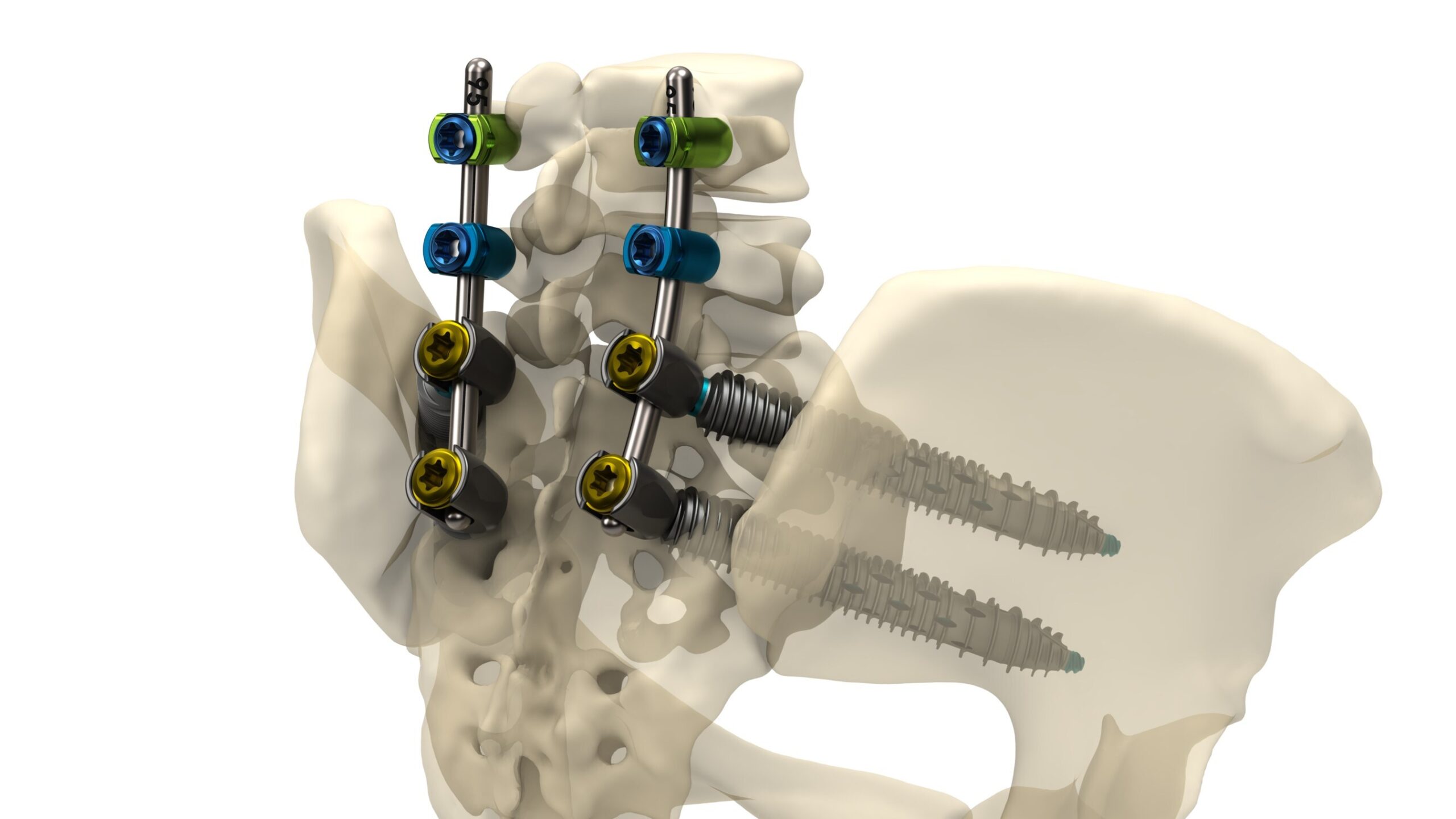
Inaugural case performed by spine surgeon Dr. Scott L. Parker highlights the system's strength, versatility, and precision for supporting multi-level spinal constructs
Medical Device News Magazine is committed to delivering high‑quality, in‑depth medical device news and biotechnology updates that include expert bylines. Our coverage offers a comprehensive view of the trends, innovations, and breakthroughs shaping the future of medical technology.
We also strive to provide a distinctive perspective on the rapidly evolving healthcare landscape. By staying connected with our reporting, you gain access to exclusive insights on industry developments, emerging technologies, and global regulatory changes that influence the medical device sector. At the same time, you join a vibrant community of professionals, innovators, and thought leaders dedicated to advancing healthcare through cutting‑edge solutions.
We invite you to explore our wide range of content—from the latest product announcements to detailed analyses of industry trends. And if our mission aligns with your goals, we offer opportunities for advertising and collaboration, helping you connect with a highly engaged audience of medical technology professionals.
Definition medical devices.
Medical Device Industry News | Latest Developments, Breakthroughs & Future Outlook
Latest News and Developments
Cartessa Aesthetics and Classys Introduce QUADESSY, a Faster, Safer and More Versatile Microneedling Radiofrequency Platform
Quadessy delivers multi-layer skin remodeling through a unique tip and dual-depth design
Medtronic and Merit Medical Systems enter into agreement to offer new, FDA-cleared ViaVerte™ system, a basivertebral nerve ablation treatment for chronic vertebrogenic lower-back pain
Medtronic's latest partnership with Merit Medical Systems introduces the groundbreaking ViaVerte™ BVNA system—an innovative solution for chronic vertebrogenic pain
Clinical Trials
Complement Therapeutics Announces First Patient Dosed with CTx001 in the Phase I/II Opti-GAIN Study for Geographic Atrophy Secondary to AMD
First patient dosed in Opti-GAIN, a first-in-human Phase I/II clinical trial of CTx001 in Geographic Atrophy secondary to AMD
Innovative Medical Equipment, LLC Highlights Published VA Clinical Study on Thermal Therapy and Pain Treatment Utilization
The study, published in The March 2026 edition of Federal Practitioner, reported an association between thermal therapy use and reduced opioid and benzodiazepine utilization, as well as lower healthcare utilization in the patient population.
Results from PARADIGM Phase 2b Trial of PrimeC in ALS Demonstrating Meaningful Clinical Outcomes and Biological Activity Published in JAMA Neurology
The Journal of the American Medical Association (JAMA Neurology) publication highlights consistent clinical and biomarker findings from NeuroSense PARADIGM Phase 2b trial in ALS, including slower functional decline, reduced risk of ALS-related complications, and modulation of disease-relevant biomarkers
Biotechnology News
iXCells Biotechnologies Joins International Collaboration to Evaluate Chemotherapeutic Toxicity in Human Organoids
Joint initiative with Rosebud Biosciences, Kantify, and funded by Incite will identify and quantify off-target toxicities in chemotherapy patients
Primerdesign Partners with Origin Sciences to Create Novel Rectal Mucosal Extraction Workflow
Robust DNA extraction supports earlier detection and diagnosis of colorectal cancer and other gastrointestinal diseases
AnalytiChem Launches New Legionella Testing Selective Agar Plate
Redipor® Legionella agar plate range - specifically designed to support the detection and enumeration of Legionella in water samples
Mergers & Acquisitions
Funding
Phantom Neuro Secures $19M Series A Funding
Investment will accelerate clinical trials and commercialization of Phantom X, with Ottobock joining Phantom Neuro’s Board of Directors
Xeltis Successfully Passes Evaluation Phase for €10Mil Investment from European Innovation Council
Funding to support commercialization of its groundbreaking bioresorbable vascular conduits notes Xeltis.
Industry Expert Bylines
5 Cardiovascular SaMD Cleared in 2023 | By Randy Horton, Chief Solutions Officer, Orthogonal
Mr. Horton advises this article takes a quick look at five interesting SaMD from the Cardiovascular panel that were cleared by the FDA in 2023. The common thread among these recently cleared SaMD is a focus on remotely monitoring patients and alerting them or their clinicians when irregular heart rhythms or dangerous heart events occur.

How Applied Research is Transforming Medical Device Startups | By Hannah Eherenfeldt, Co-founder & CEO of ReSuture
Medical device startups face numerous barriers when trying to penetrate the crowded startup landscape. Fundraising is highly competitive, and many products fail to reach the market due to insufficient funding for research, validation, and testing. The applied research model provides a solution.This industry expert notes this is where the applied research model comes in.

Commercial Success In Global Expansion | Why Compliance and Regulation Don’t Have to Be So Hard | By Phyllis Meng, co-founder and CEO of Pure Global
Do compliance and regulation have to be so challenging? Our expert shares their views.
FDA Medical Device Updates
Market Reports
restor3d Receives FDA Clearance for iTotal IdentityTM CR 3DP Porous Fully Personalized Total Knee Replacement System
This milestone marks a significant achievement in expanding restor3d’s knee replacement technology since the acquisition of Conformis, introducing the first cementless offering to the patient-specific implant portfolio. The company plans a limited market release in Q3 of 2025.
Phantom Neuro Receives FDA Breakthrough Device & TAP Designations, Solidifying Position as a Neurotech Leader
“Receiving both of these certified designations from the FDA is a tremendous validation of our work,” said Dr. Connor Glass, Founder and CEO of Phantom Neuro. “These recognitions validate our technology and reflect our commitment to creating scalable, real-world solutions that restore functionality and independence to amputees and those with functional disabilities. Our goal is to bring Phantom X to patients faster, and achievements like this accelerate our clinical and regulatory processes.”
Executives
Dan Cummings Is Appointed Chief Commercial Officer of Precision AQ
Dan Cummings will be responsible for Precision AQ’s commercial and go-to-market strategies, with the goal of bringing Precision AQ’s comprehensive suite of capabilities to help biopharmaceutical companies advance life-changing therapies to market.
Boston Scientific Chief Financial Officer Dan Brennan to Retire | Jon Monson, SR VP, Investor Relations to succeed Brennan
Brennan will transition out of the chief financial officer position at the end of June and is expected to remain with Boston Scientific as a senior advisor through early October 2025.

New Executive Appointments Announced Avania
“We recognize the MedTech industry is consistently changing and remain committed to providing flexible solution-driven partnerships that guide our clients from concept to commercialization. Our ability to navigate the changes in the industry is rooted in the strength of our team,” said Jason Monteleone, president and CEO of Avania.
Advancements In Imaging
Carestream Health Earned 70 New Patents In 2024
Carestream Health notes twenty-seven of the patents were awarded by the U.S. Patent and Trademark Office; an additional 43 patents were received in European and Asian countries.
Insightec MR-Guided Focused Ultrasound Technology Recommended for National Coverage for Dutch Patients Living with Essential Tremor
According to an Insightec study, patients experienced significant reductions in upper limb tremor, tremor-related disability, and quality of life. The most common intraprocedural adverse events included dizziness, headache, nausea, and vomiting, which all resolved. Post-procedural adverse events included paraesthesia and gait disturbance, which were often transient or resolved within 12 months.
![IGT IVUS[18] - Medical Device News Magazine IVUS](https://infomeddnews.com/wp-content/uploads/2024/01/IGT-IVUS18-400x267.jpg)
Multidisciplinary Panel of Experts from Prominent Cardiovascular Societies Advocate for Broader Adoption of IVUS in Peripheral Interventions to Improve Patient Care
Chris Landon, Business Leader Image Guided Therapy Devices at Philips:
“IVUS is a technology that has demonstrated an ability to both support safer procedures for patients and procedural staff alike including reductions in radiation exposure. As the global leader in IVUS solutions, we are grateful for the rigorous multidisciplinary approach to developing expert consensus on the value of IVUS and we look forward to working with all stakeholders to standardize the use of IVUS and reduce the barriers preventing utilization.”
Hospitals In the News
Non-Profit News
Healthcare: Understanding the Business Side of the Industry and Its Implications

Medical Devices In Clinical Mental Health Counseling
The advent of medical devices in counseling has revolutionized the provision of mental health services with well-tailored approaches, ultimately enhancing patients’ experiences. This is a wake-up call for all mental health professionals to accept these technological changes, continuously learn, and take advantage of medical devices to advance their practices and improve the lives of their patients.

4 Key Reasons to Embrace EMR for Mental Health Specialists
As a dedicated mental health specialist, you always strive hard to help individuals navigate the complexities of their mental diseases. However, the nature of mental disorders is constantly evolving, compelling you to adapt to the change.

Top 5 Patient Engagement Platforms Revolutionizing Healthcare
Patient engagement platforms are changing how healthcare is delivered by improving communication and handling administrative tasks. Read on.

Unveiling the Hidden Artistry: How Data Annotation Transforms Medical Imaging into AI Masterpieces
In the grand tapestry of modern medicine, artificial intelligence is weaving threads that are redefining diagnostics and patient care. But behind the scenes of every groundbreaking AI model lies a silent artist: the meticulous craft of data annotation. This unsung hero transforms ordinary medical images and videos into rich, informative datasets that teach AI how to “see” like a seasoned physician.