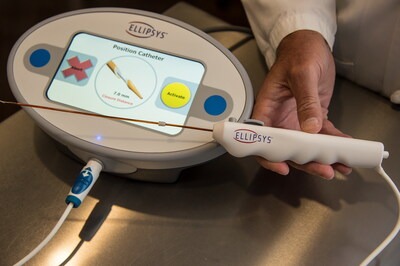
Ellipsys Medical, Inc. today announced that the Ellipsys® Vascular Access System will once again be commercially available, with initial availability anticipated in mid-2026. The announcement marks the planned reintroduction of the minimally invasive technology to clinicians and patients following a period of limited market availability. The Ellipsys System, previously available...
Medical Device News Magazine is committed to delivering high‑quality, in‑depth medical device news and biotechnology updates that include expert bylines. Our coverage offers a comprehensive view of the trends, innovations, and breakthroughs shaping the future of medical technology.
We also strive to provide a distinctive perspective on the rapidly evolving healthcare landscape. By staying connected with our reporting, you gain access to exclusive insights on industry developments, emerging technologies, and global regulatory changes that influence the medical device sector. At the same time, you join a vibrant community of professionals, innovators, and thought leaders dedicated to advancing healthcare through cutting‑edge solutions.
We invite you to explore our wide range of content—from the latest product announcements to detailed analyses of industry trends. And if our mission aligns with your goals, we offer opportunities for advertising and collaboration, helping you connect with a highly engaged audience of medical technology professionals.
Definition medical devices.
Medical Device Industry News | Latest Developments, Breakthroughs & Future Outlook
Latest News and Developments
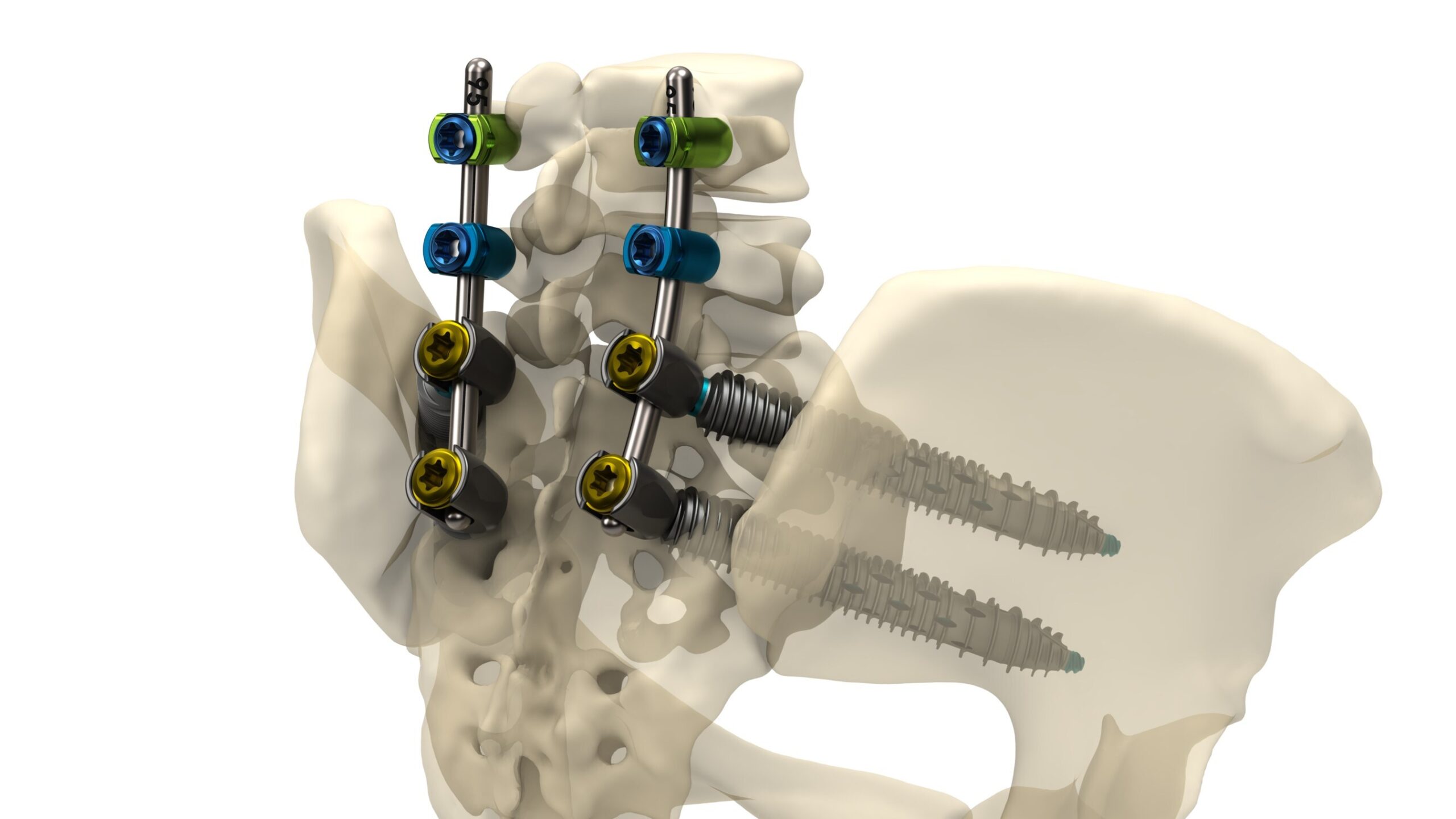
Genesys Spine Announces Launch of the SIros-X™ System for Sacroiliac Joint Fusion and Pelvic Fixation of Pedicle Screw Constructs
Inaugural case performed by spine surgeon Dr. Scott L. Parker highlights the system's strength, versatility, and precision for supporting multi-level spinal constructs
Cartessa Aesthetics and Classys Introduce QUADESSY, a Faster, Safer and More Versatile Microneedling Radiofrequency Platform
Quadessy delivers multi-layer skin remodeling through a unique tip and dual-depth design
Clinical Trials
Complement Therapeutics Announces First Patient Dosed with CTx001 in the Phase I/II Opti-GAIN Study for Geographic Atrophy Secondary to AMD
First patient dosed in Opti-GAIN, a first-in-human Phase I/II clinical trial of CTx001 in Geographic Atrophy secondary to AMD
Innovative Medical Equipment, LLC Highlights Published VA Clinical Study on Thermal Therapy and Pain Treatment Utilization
The study, published in The March 2026 edition of Federal Practitioner, reported an association between thermal therapy use and reduced opioid and benzodiazepine utilization, as well as lower healthcare utilization in the patient population.
Results from PARADIGM Phase 2b Trial of PrimeC in ALS Demonstrating Meaningful Clinical Outcomes and Biological Activity Published in JAMA Neurology
The Journal of the American Medical Association (JAMA Neurology) publication highlights consistent clinical and biomarker findings from NeuroSense PARADIGM Phase 2b trial in ALS, including slower functional decline, reduced risk of ALS-related complications, and modulation of disease-relevant biomarkers
Biotechnology News
Transgene and NEC Bio Sign License Agreement to Prepare the Next Steps of the Development of TG4050 in Head and Neck Cancer
Akira Kitamura, GM, AI Drug Development Division of NEC Corporation and CEO of NEC Bio, added, "This agreement is an important milestone in our partnership with Transgene and reflects NEC’s long-term commitment to the development of TG4050
iXCells Biotechnologies Joins International Collaboration to Evaluate Chemotherapeutic Toxicity in Human Organoids
Joint initiative with Rosebud Biosciences, Kantify, and funded by Incite will identify and quantify off-target toxicities in chemotherapy patients
Primerdesign Partners with Origin Sciences to Create Novel Rectal Mucosal Extraction Workflow
Robust DNA extraction supports earlier detection and diagnosis of colorectal cancer and other gastrointestinal diseases
Mergers & Acquisitions
Funding
Enifer Secures €36M In Funding
Enifer, a biotech scaleup is gearing up the production of its PEKILO® mycoprotein with a new €33M factory located in Kirkkonummi, Finland. With already existing collaborations with the world’s leading food and feed companies, Enifer is prepared to annually upcycle food industry side streams into up to 3,000 tons of its sustainable fungi-based protein
Phenomix Sciences Raises $7.8M in Funding to Accelerate Adoption of Obesity Precision Medicine
Funding will accelerate the commercialization and adoption of the MyPhenome™ test in the obesity specialist market and the wider industry. The MyPhenome test equips physicians with essential tools to develop effective treatment plans including lifestyle and diet intervention as well as medication and/or procedure recommendations reports Phenomix Sciences.
Industry Expert Bylines

How Medical Professionals Can Solve Healthcares Medical Waste Problem | By Steven John Cumper, B.App.Sc. (Osteo.)
In this article, Cumper evaluates some of the most common causes of medical waste, the challenges of managing them, and how to reduce waste throughout the entire industry. Yes, we have a medical waste problem!

It is Time to Optimize EHR Systems and Experiences around Patient Needs | Raghid El-Yafouri, Digital Transformation Strategist at Bottle Rocket
Raghid El-Yafouri, Digital Transformation Strategist at Bottle Rocket, delves into the possibilities of optimizing EHR systems around patient preferences, transforming healthcare into a truly patient-centered experience.

AR + AI in MedTech: Innovating Healthcare and Expanding Verticals | By Michael Freeman, CEO of Ocutrx Technologies Inc
Ocutrx Technologies, Inc. is a future-forward medical device company, harnessing the power of both AI and AI for patients and surgeons. Read on to learn more.
FDA Medical Device Updates
Market Reports
Executives
Advancements In Imaging
GE Healthcare’s AIR Recon DL Receives FDA Clearance of 3D & Motion-Insensitive Imaging Applications for Next-Level Image Quality and Patient Experience in MRI
GE Healthcare advises the benefits of AIR Recon DL are extended by these new features to nearly all Magnetic Resonance Imaging (MRI) clinical procedures, covering all anatomies and enabling better image quality, shorter scan times, and enhanced patient experience.

Mobile Imaging Became a Hero for This Small Central NY Hospital – During and After Peak COVID-19
Oswego Hospital’s innovative imaging staff maximized its use of the CARESTREAM DRX-Revolution Mobile Imaging System to care for patients
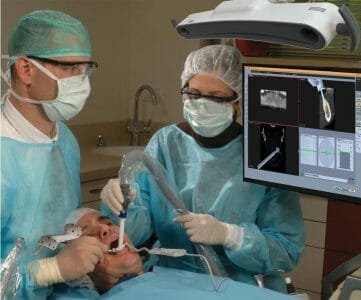
Image Navigation to Launch Newest Image Guided Implantology Technology at the European Academy of Osseointegration Congress in Geneva
Image Navigation Ltd. will be exhibiting the new IGI 2.0 at the European Academy of Osseointegration Congress, at Palexpo Geneva, Switzerland, September 29 – October 1, Booth H31.
Hospitals In the News
Non-Profit News
Healthcare: Understanding the Business Side of the Industry and Its Implications

Top VoIP Technology Challenges For Healthcare Industry
Voice over Internet Protocol (VoIP) technology has revolutionized communication in various industries, including healthcare. With its cost-effectiveness, ease of use, and flexibility, VoIP has become

Best Practices For Automation In Healthcare Sector
In the rapidly evolving landscape of the healthcare sector, technological advancements have become a driving force behind increased efficiency and improved patient care. One such
The Power Of Instagram Reels: A Guide For Health Care Businesses
Instagram Reels have taken the world by storm. Offering an incredible opportunity for companies to connect with customers and fans on an emotional level, Reels
Precision Medicine Implications: An Analysis for Executive Management
Precision medicine has heralded a new era in healthcare, marking a shift from a one-size-fits-all approach to personalized, patient-specific care. At its core, precision medicine