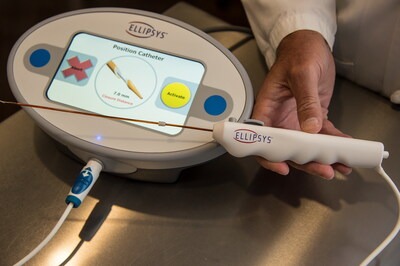
Ellipsys Medical, Inc. today announced that the Ellipsys® Vascular Access System will once again be commercially available, with initial availability anticipated in mid-2026. The announcement marks the planned reintroduction of the minimally invasive technology to clinicians and patients following a period of limited market availability. The Ellipsys System, previously available...
Medical Device News Magazine is committed to delivering high‑quality, in‑depth medical device news and biotechnology updates that include expert bylines. Our coverage offers a comprehensive view of the trends, innovations, and breakthroughs shaping the future of medical technology.
We also strive to provide a distinctive perspective on the rapidly evolving healthcare landscape. By staying connected with our reporting, you gain access to exclusive insights on industry developments, emerging technologies, and global regulatory changes that influence the medical device sector. At the same time, you join a vibrant community of professionals, innovators, and thought leaders dedicated to advancing healthcare through cutting‑edge solutions.
We invite you to explore our wide range of content—from the latest product announcements to detailed analyses of industry trends. And if our mission aligns with your goals, we offer opportunities for advertising and collaboration, helping you connect with a highly engaged audience of medical technology professionals.
Definition medical devices.
Medical Device Industry News | Latest Developments, Breakthroughs & Future Outlook
Latest News and Developments
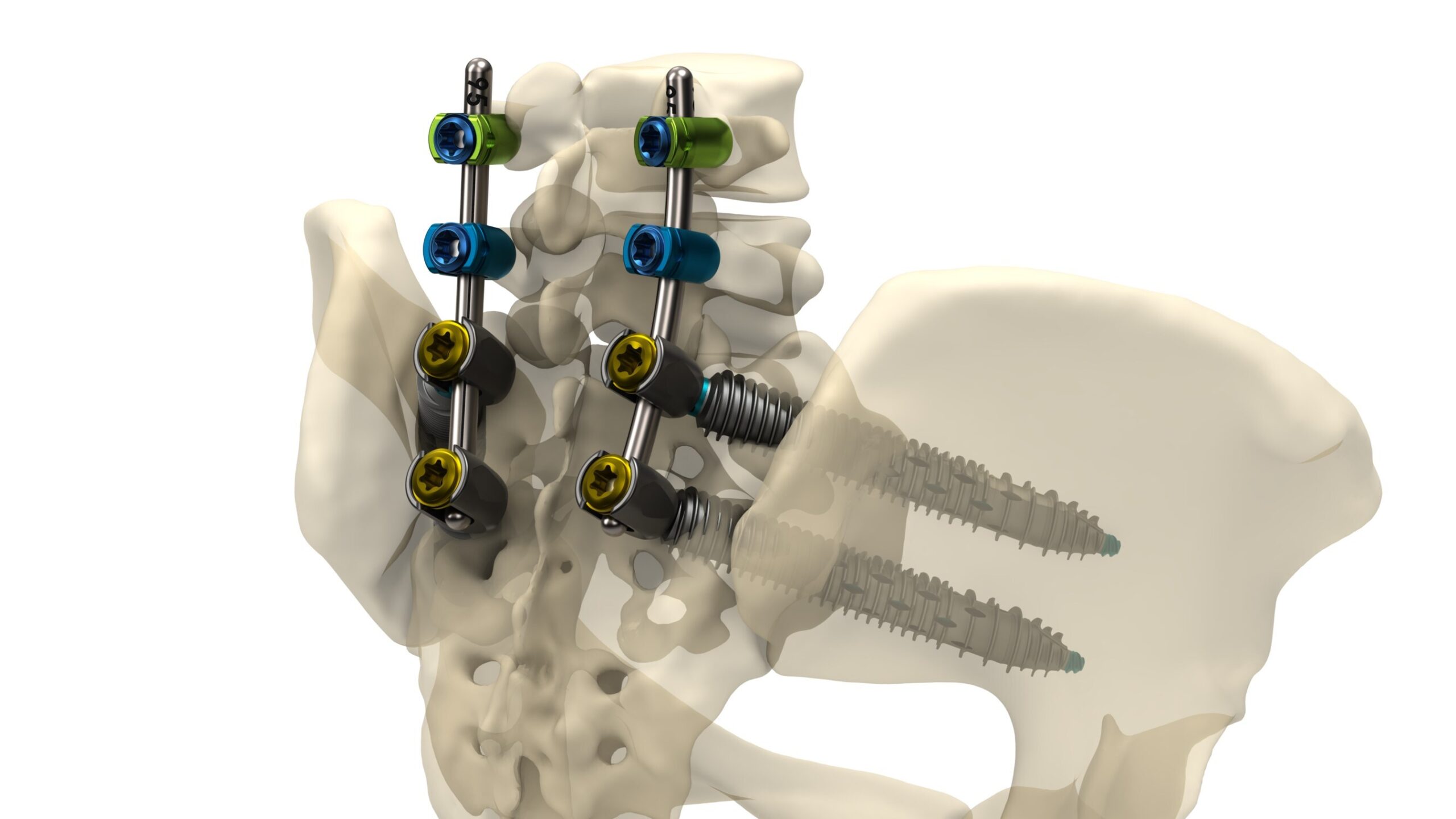
Genesys Spine Announces Launch of the SIros-X™ System for Sacroiliac Joint Fusion and Pelvic Fixation of Pedicle Screw Constructs
Inaugural case performed by spine surgeon Dr. Scott L. Parker highlights the system's strength, versatility, and precision for supporting multi-level spinal constructs
Cartessa Aesthetics and Classys Introduce QUADESSY, a Faster, Safer and More Versatile Microneedling Radiofrequency Platform
Quadessy delivers multi-layer skin remodeling through a unique tip and dual-depth design
Clinical Trials
Medicus Pharma Submits Optimized Phase 2 Study Protocol to U.S. FDA for Teverelix in Acute Urinary Retention
Mechanism-Driven Study Design Focused on Capital Efficiency and Accelerated Development for Near-Term Value Creation, Addressing a $2 Billion Potential Target Market
Complement Therapeutics Announces First Patient Dosed with CTx001 in the Phase I/II Opti-GAIN Study for Geographic Atrophy Secondary to AMD
First patient dosed in Opti-GAIN, a first-in-human Phase I/II clinical trial of CTx001 in Geographic Atrophy secondary to AMD
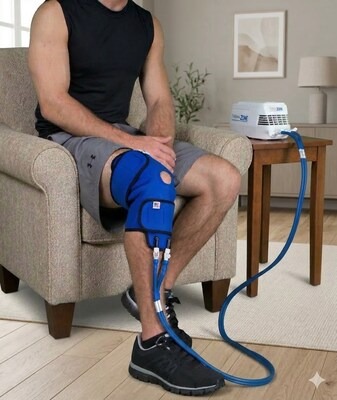
Innovative Medical Equipment, LLC Highlights Published VA Clinical Study on Thermal Therapy and Pain Treatment Utilization
The study, published in The March 2026 edition of Federal Practitioner, reported an association between thermal therapy use and reduced opioid and benzodiazepine utilization, as well as lower healthcare utilization in the patient population.
Biotechnology News
Variational AI Releases Enki 4: Major Update to Foundation Model for Small-Molecule Drug Discovery
Variational AI introduces Enki™ 4: an improved algorithm and architecture designed to scale, expanding the pre-trained target coverage from 592 to 760 and now applicable to proximity-based therapeutics and novel payloads for antibody drug conjugates
Transgene and NEC Bio Sign License Agreement to Prepare the Next Steps of the Development of TG4050 in Head and Neck Cancer
Akira Kitamura, GM, AI Drug Development Division of NEC Corporation and CEO of NEC Bio, added, "This agreement is an important milestone in our partnership with Transgene and reflects NEC’s long-term commitment to the development of TG4050
iXCells Biotechnologies Joins International Collaboration to Evaluate Chemotherapeutic Toxicity in Human Organoids
Joint initiative with Rosebud Biosciences, Kantify, and funded by Incite will identify and quantify off-target toxicities in chemotherapy patients
Mergers & Acquisitions
Funding

Conformal Medical Raises $35 Million in Oversubscribed Series D Round
The highly supported round reflects the progress and achievements made since its last funding round, which includes the initiation of the CONFORM Clinical Trial. The CONFORM Trial is the company’s pivotal trial evaluating the safety and efficacy of the CLAAS® System compared to other commercially available LAAO devices and will support U.S. Food and Drug Administration (FDA) pre-market approval.
PathMaker Neurosystems Receives $200,000 START Award from MassVentures
“We are grateful for the Stage 2 award from MassVentures as we work to bring MyoRegulator® to market,” said Nader Yaghoubi, M.D., Ph.D., Co-Founder and Chief Executive Officer of PathMaker. “As we continue our US multi-center pivotal trial in post-stroke spasticity and work to launch our first clinical trial in ALS this year, MassVentures support for late-stage development of our product is greatly appreciated.”
Industry Expert Bylines

RTS Tactical Rapid Deploy IFAK By Mendel Berns
Mendel Berns writes, “When treating an injury in the field, time is critical. Every second counts. You need to be ready when called upon to save the life of a team member or perhaps your own.” Read what he has to say about having the best medical equipment.

Patient Payments: Top 3 Reasons Healthcare Practices Are Turning to Technology
d’Artagnan Osborne writes, “Technology has come a long way to help improve the patient payment process.” He describes how adopting new technology can benefit a healthcare practice and the patient! Read on to learn more.

The Hospital at Home: A New Approach to Patient Care – By Stuart Long, CEO of InfoBionic
Mr. Long writes, “The Hospital at Home model of healthcare gives patients more freedom of choice and autonomy over their own health and treatment decisions.” Read his expert views.
FDA Medical Device Updates
Market Reports
Executives
Advancements In Imaging
Hospitals In the News
Non-Profit News
Healthcare: Understanding the Business Side of the Industry and Its Implications
Healthcare Software Testing: Launch Plan & Key Cost Factors
Healthcare software testing enables the adoption of necessary protocols for data interchange, ensures that client statistics are secure, and ensures that medical IT systems operate

Starting Your Own Functional Medicine Practice: A Guide for Doctors Who Do It All
According to the Centers for Disease Control (CDC) the use of nutrient boosts is increasing in the US. Also, the number of Americans using dietary
Make Your Recruitment Process Run More Smoothly With These Tips
Recruitment Process Looking to hire new employees but feeling overwhelmed? Relax! The recruitment process can seem daunting, but it can run more smoothly with the

The Role and Impact of Public Health Practitioners Today
The public health system in the United States is a complex network of federal, state, and local entities that work together to protect and improve




![Is Nail Fungus a Health Concern_ (1)[1]-1 - Medical Device News Magazine Nail Fungus](https://infomeddnews.com/wp-content/uploads/2024/09/Is-Nail-Fungus-a-Health-Concern_-11-1-1200x795.jpg)

![image[1] - Medical Device News Magazine Financial Stability](https://infomeddnews.com/wp-content/uploads/2023/06/image1-1-400x266.jpg)

