Abstract
Human physiology is regulated by circadian rhythms that coordinate metabolism, hormone secretion, mitochondrial function, and cellular repair across the 24-hour cycle. Despite this temporal organization, nutritional interventions are commonly administered without consideration of biological timing. Increasing evidence indicates that metabolic responses to nutrient intake vary according to circadian phase, influencing glucose handling, energy metabolism, nutrient absorption, and recovery processes. This article examines the mechanistic relationship between circadian regulation and metabolic function, with emphasis on nutrient timing, transporter activity, and mitochondrial rhythmicity. A phase-based nutritional model organized around activation, metabolic regulation, and recovery is discussed as a physiologically aligned approach to daily supplementation. Collectively, current findings support the concept that both nutrient composition and timing contribute to metabolic outcomes and should be considered in the design of nutritional strategies.
- Introduction
Nutritional science has traditionally focused on the composition of dietary intake, emphasizing macronutrients, micronutrients, and bioactive compounds as primary determinants of metabolic function. In contrast, the timing of nutrient exposure has received comparatively limited attention. However, accumulating evidence indicates that human metabolism is not constant throughout the day, but instead follows pronounced circadian patterns regulated by endogenous biological clocks.
Circadian rhythms influence a broad range of physiological processes, including glucose metabolism, mitochondrial activity, hormonal signaling, sleep–wake cycles, and cellular repair. These rhythms are coordinated by a central pacemaker located in the suprachiasmatic nucleus (SCN) of the hypothalamus and synchronized with peripheral clocks present in metabolic tissues such as the liver, skeletal muscle, adipose tissue, pancreas, and gastrointestinal tract (Takahashi, 2017; Bass & Takahashi, 2010). Together, these systems temporally organize metabolic activity in anticipation of recurring cycles of feeding, fasting, activity, and rest.
As illustrated in Figure 1, metabolic and hormonal processes exhibit distinct temporal patterns across the 24-hour cycle. Glucose tolerance, mitochondrial function, cortisol secretion, and recovery-related pathways vary according to circadian phase, indicating that nutrient utilization is closely linked to biological timing.
Despite this temporal organization, most nutritional interventions and dietary supplements are administered uniformly without regard to circadian physiology. This discrepancy raises an important question: can nutritional strategies be improved by aligning nutrient delivery with the temporal organization of metabolism?
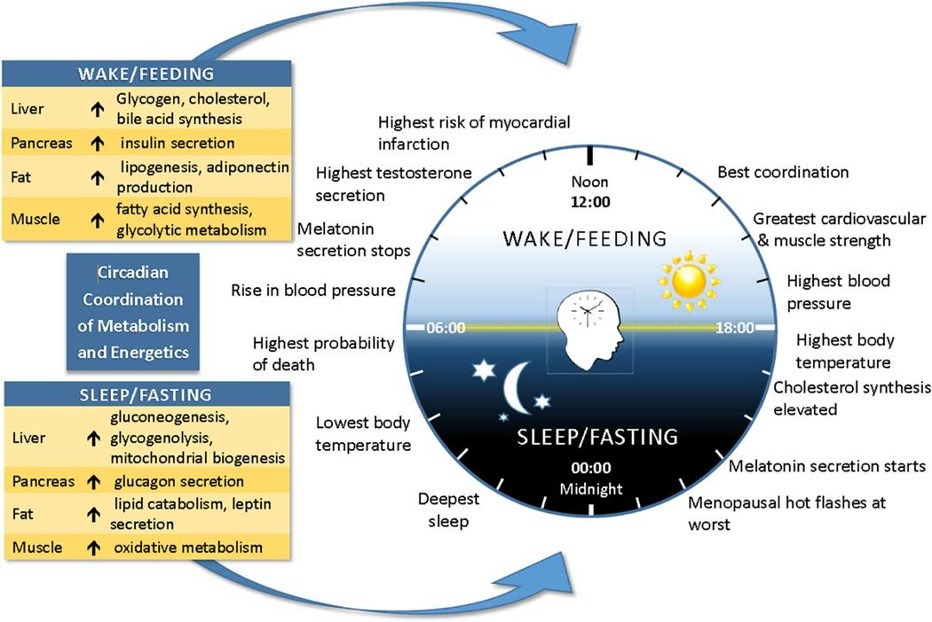
Figure 1. Circadian organization of metabolic physiology across the 24-hour cycle.
Circadian rhythms coordinate temporal variation in hormonal signaling, glucose metabolism, mitochondrial activity, nutrient utilization, and recovery-related processes. Representative patterns of cortisol secretion, insulin sensitivity, metabolic activity, and rest-activity transitions are illustrated across the biological day and night.
- Circadian Regulation of Metabolism
2.1 Central and Peripheral Clock Systems
The circadian system operates through a hierarchical network of transcriptional–translational feedback loops that generate approximately 24-hour oscillations in gene expression and cellular function. The central pacemaker located in the suprachiasmatic nucleus (SCN) of the hypothalamus is primarily entrained by environmental light–dark cycles and synchronizes peripheral clocks throughout the body. Peripheral clocks are present in metabolically active tissues including the liver, skeletal muscle, adipose tissue, pancreas, and gastrointestinal tract, where they regulate tissue-specific processes involved in energy homeostasis and substrate utilization (Bass & Takahashi, 2010).
At the molecular level, circadian timing is governed by core clock genes organized into self-regulating feedback loops. CLOCK and BMAL1 transcription factors activate expression of PER and CRY genes, whose protein products subsequently inhibit CLOCK–BMAL1 activity, thereby generating rhythmic oscillations in gene expression (Panda, 2016). These molecular clocks regulate a broad range of metabolic pathways, including glucose transport, lipid metabolism, and mitochondrial activity.
As illustrated in Figure 2, the SCN coordinates peripheral clocks that regulate tissue-specific metabolic functions. The liver clock influences gluconeogenesis and glucose export, skeletal muscle clocks regulate mitochondrial respiration and insulin sensitivity, while intestinal clocks affect nutrient absorption and transporter expression. Adipose and pancreatic clocks further contribute to metabolic regulation through effects on lipid handling, insulin secretion, and energy storage.
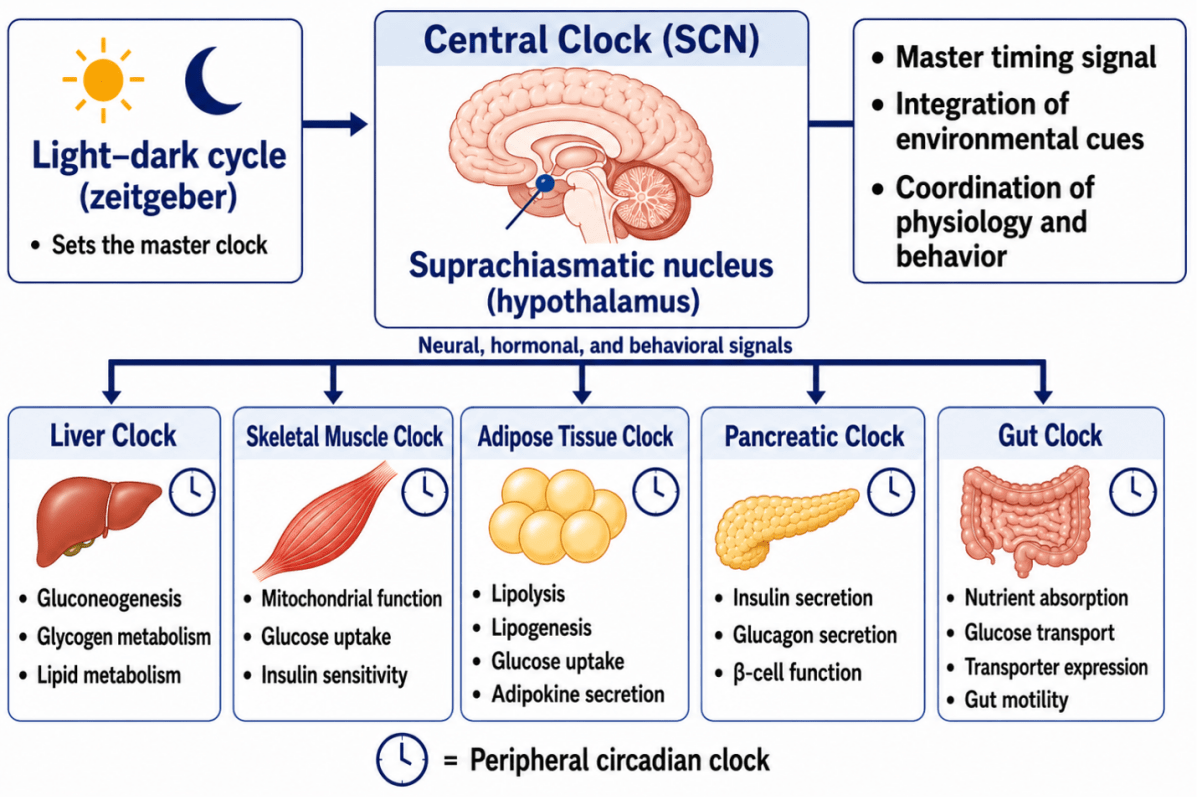
Figure 2. Central and Peripheral Circadian Clocks Regulating Metabolic Function
The suprachiasmatic nucleus (SCN) acts as the central circadian pacemaker and synchronizes peripheral clocks in metabolic tissues including liver, skeletal muscle, adipose tissue, pancreas, and gastrointestinal tract. These tissue-specific oscillators regulate glucose metabolism, mitochondrial activity, nutrient absorption, insulin signaling, and energy homeostasis across the 24-hour cycle.
Optimal physiological homeostasis, including metabolism and sleep-wake regulation, relies on synchronized central and peripheral circadian clocks to prevent accelerated aging. While the SCN provides the primary timing signal, peripheral clocks are also responsive to feeding-fasting cycles and nutrient exposure. Disruption of this synchronization, such as through irregular eating patterns, sleep disturbance, or shift work, can impair metabolic regulation and contribute to circadian misalignment.
These mechanisms demonstrate that circadian organization is an active regulatory system integrating environmental cues with metabolic physiology, providing a mechanistic basis for the role of nutrient timing in metabolic function.
2.2 Temporal Organization of Energy Metabolism
Metabolic processes are organized into distinct temporal phases under circadian regulation, reflecting coordinated changes in hormonal signaling, autonomic balance, and energy demand. Across the 24-hour cycle, the body transitions through periods of activation, nutrient processing, and recovery, each associated with specific metabolic priorities.
The morning phase is characterized by increased cortisol secretion and sympathetic activity, promoting glucose mobilization, hepatic output, and mitochondrial readiness following the overnight fast. These coordinated responses support energy production, cognitive alertness, and preparation for daytime activity.
During the daytime phase, nutrient intake and metabolic processing predominate. Insulin sensitivity is generally higher, supporting efficient glucose uptake and substrate utilization. Digestive and absorptive processes are also optimized during this period, facilitating integration of dietary inputs into energy metabolism and maintenance of redox balance.
In contrast, the night phase reflects a shift toward parasympathetic dominance and reduced energy expenditure. As melatonin levels rise and cortisol declines, metabolism transitions toward cellular repair and recovery. Processes such as cellular restoration, regeneration, and detoxification become more prominent, while mitochondrial activity shifts from maximal ATP production toward maintenance functions.
As illustrated in Figure 3, glucose tolerance, lipid metabolism, and energy expenditure exhibit circadian variation across the day. Glucose handling is typically more efficient earlier in the active phase, whereas impaired tolerance is commonly observed during the biological night or under conditions of circadian misalignment (Morris et al., 2015).
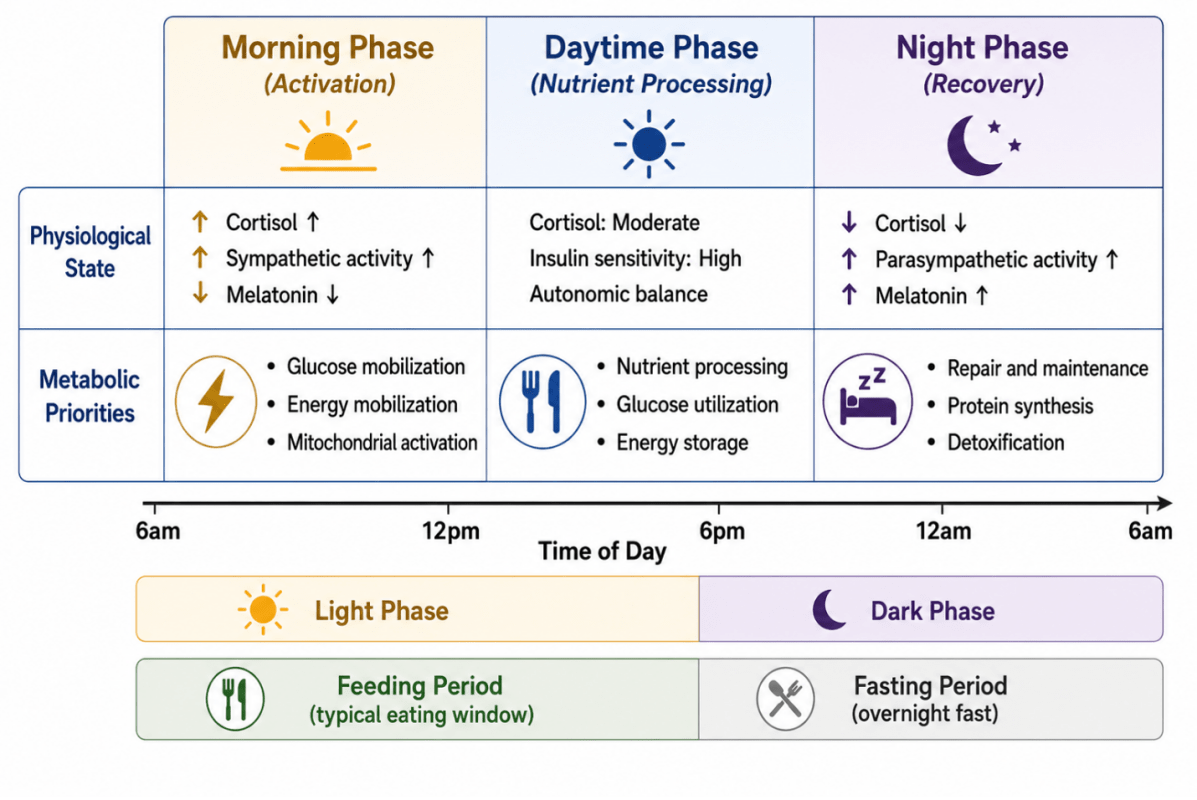
Figure 3. Temporal Organization of Metabolic Phases Across the 24-Hour Cycle
Circadian rhythms coordinate predictable transitions between activation, nutrient processing, and recovery phases across the biological day. These temporal changes influence hormonal signaling, mitochondrial activity, glucose metabolism, nutrient utilization, and cellular repair processes.
These observations indicate that metabolic efficiency is closely linked to biological timing. Recognizing the temporal organization of energy metabolism provides a physiological basis for aligning nutritional inputs with circadian phase and metabolic demand.
2.3 Mitochondrial Rhythmicity
Mitochondrial function is regulated by circadian rhythms and varies across the 24-hour cycle. Processes such as oxidative phosphorylation and substrate utilization are coordinated with the rest-activity rhythm, reflecting the integration of molecular clock mechanisms with cellular energy metabolism.
Experimental studies have shown that mitochondrial oxidative capacity, ATP production, and reactive oxygen species generation fluctuate in a time-dependent manner. These oscillations are driven by circadian control of genes involved in mitochondrial biogenesis and respiratory function, as well as by regulation of cofactors such as NAD⁺ that influence oxidative metabolism (Peek et al., 2013).
This rhythmicity allows mitochondria to adapt to changing physiological demands. During active periods, mitochondrial pathways support efficient ATP production, while during rest phases they shift toward repair and regeneration, including processes such as mitophagy. Disruption of circadian rhythms can impair these functions, leading to reduced metabolic flexibility, increased inflammation, and accelerated aging.
Mitochondrial efficiency therefore varies according to circadian phase rather than remaining constant throughout the day. Circadian modulation of mitochondrial processes provides a mechanistic connection between environmental cues and cellular energy metabolism, highlighting the importance of temporal factors in metabolic regulation.
- Nutrient Timing and Metabolic Outcomes
3.1 Circadian Variation in Glucose Metabolism
Glucose metabolism is strongly influenced by circadian regulation, with measurable variation in insulin sensitivity, glucose tolerance, and substrate utilization across the 24-hour cycle. These changes are coordinated by central and peripheral clocks that regulate genes involved in glucose transport, insulin signaling, and hepatic glucose production. Consequently, glycemic control depends not only on nutrient composition but also on the timing of intake relative to circadian phase.
Under typical physiological conditions, insulin sensitivity is generally higher earlier in the day, supporting efficient glucose uptake in peripheral tissues and stable metabolic control. In contrast, insulin responsiveness declines later in the day, and glucose disposal becomes less efficient. These shifts are associated with circadian variation in hormonal signaling, including fluctuations in cortisol and melatonin that influence metabolic pathways and substrate utilization.
Human studies have demonstrated that meal timing significantly affects glucose metabolism. Food intake during the biological day is commonly associated with improved glucose tolerance and reduced postprandial glycemic responses. Conversely, food consumption during the biological night or under conditions of circadian misalignment can impair glucose handling and reduce insulin sensitivity (Scheer et al., 2009; Morris et al., 2015).
At the molecular level, circadian clocks regulate metabolic pathways through control of glucose transporters, enzymes involved in glycolysis and gluconeogenesis, and components of insulin signaling. These regulatory mechanisms help align glucose utilization with anticipated energy demand during the active phase of the circadian cycle.
Current evidence supports the view that nutrient timing contributes independently to metabolic outcomes. Aligning carbohydrate intake with periods of greater insulin sensitivity may improve metabolic efficiency and glycemic regulation, supporting the broader concept of circadian-aligned nutritional strategies.
3.2 Absorption and Transport Mechanisms
Nutrient absorption is highly regulated by circadian rhythms. Absorptive capacity of the intestine changes across the day in response to coordinated regulation by circadian clocks and feeding patterns, resulting in time-dependent differences in nutrient bioavailability.
Experimental studies have demonstrated that key transporters involved in nutrient uptake exhibit diurnal rhythmicity. The sodium/glucose cotransporter SGLT1 shows variation in expression and functional activity across the circadian cycle, reflecting changes in glucose absorption (Tavakkolizadeh et al., 2001). Similarly, the peptide transporter PepT1, which mediates uptake of dipeptides and tripeptides, exhibits time-dependent activity influenced by feeding conditions (Pan et al., 2002; Pan et al., 2004). These observations suggest that intestinal nutrient uptake is dynamically regulated rather than constant throughout the day.
As illustrated in Figure 4, peptide-bound nutrients utilize transport pathways that differ from those used by many free amino acids. Dipeptides and tripeptides are primarily absorbed via PepT1, enabling efficient uptake under a broad range of physiological conditions (Daniel, 2004). In contrast, free amino acids rely on multiple transporter systems that may be subject to substrate competition. These differences in transport characteristics may influence both absorption efficiency and the kinetics of nutrient availability.
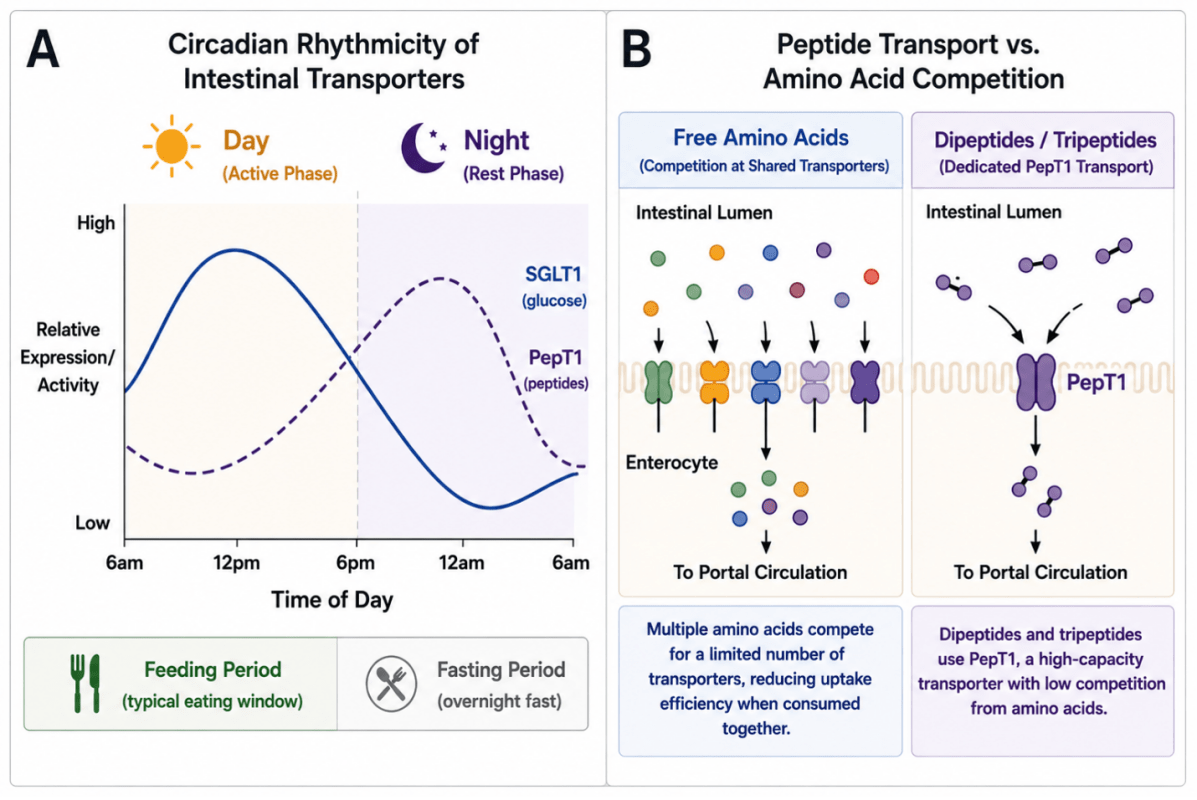
Figure 4. Transporter Rhythmicity and Peptide Uptake
These observations suggest that intestinal nutrient uptake is dynamically regulated rather than constant throughout the day.
Temporal regulation of transporter activity, together with differences in nutrient transport pathways, indicates that both nutrient form and timing contribute to bioavailability. Aligning nutrient delivery with periods of greater transporter activity and metabolic demand may therefore improve the efficiency of nutrient utilization.
3.3 Nutrient Competition and Functional Specificity
Nutrient utilization is influenced not only by availability but also by competition for shared transport systems and metabolic pathways. Many nutrients rely on common carrier mechanisms, which can limit uptake efficiency when multiple substrates are present simultaneously. A well-characterized example is the transport of large neutral amino acids across the blood–brain barrier, where substrates compete for carrier-mediated uptake, thereby influencing downstream processes such as neurotransmitter synthesis (Fernstrom, 2013).
This competitive environment highlights the importance of both nutrient form and timing. When substrates with overlapping transport requirements are consumed together, competition may reduce effective delivery to target tissues. In contrast, temporal separation of intake may help minimize competition and improve functional specificity. Within this context, short peptides represent a distinct class of substrates. Dipeptides and tripeptides are primarily absorbed via the PepT1 transporter, which operates independently of many amino acid transport systems. This separation reduces direct competition with free amino acids during intestinal uptake and may contribute to more consistent delivery under mixed nutrient conditions.
The use of peptide-bound forms therefore introduces an additional dimension to nutrient delivery by diversifying transport pathways. When combined with time-structured intake, this approach may allow for improved alignment between substrate availability, transporter activity, and metabolic demand. From a systems perspective, integrating both timing and transport specificity provides a framework for enhancing functional targeting without altering the underlying biochemical roles of individual nutrients.
- Circadian Alignment as a Design Principle
Conventional supplementation strategies are typically static, delivering multiple nutrients simultaneously without regard to biological timing. This approach assumes relatively uniform metabolic responsiveness across the day, despite evidence that processes such as glucose handling, mitochondrial activity, nutrient absorption, and hormonal signaling vary according to circadian phase. Consequently, identical nutritional inputs may produce different metabolic effects depending on when they are consumed.
A circadian-aligned strategy incorporates three interrelated principles. First, temporal organization of physiology defines dominant functional states across the day, including activation during the waking period, metabolic processing during active feeding periods, and recovery during the rest phase. Second, nutrients can be matched to these functional states according to their primary metabolic roles and transport characteristics. Third, the sequence of delivery determines when nutrients are administered relative to circadian phase, feeding cycles, and anticipated metabolic demand.
Integrating these variables shifts the emphasis from isolated ingredients toward coordinated nutritional organization. Rather than maximizing the number of compounds delivered simultaneously, this approach prioritizes alignment between nutrient function, transport dynamics, and metabolic state. Within this context, both nutrient form and delivery sequence may influence utilization efficiency. Peptide-bound substrates, for example, can diversify intestinal transport pathways, while temporal separation of intake may reduce substrate competition and improve functional specificity.
Viewed from a systems perspective, circadian alignment provides a structured method for organizing nutritional inputs across the 24-hour cycle. This model does not alter the intrinsic biochemical properties of individual nutrients, but instead seeks to improve utilization efficiency by coordinating delivery with the temporal organization of metabolism.
- A Phase-Based Model of Daily Nutrition
A circadian-aligned nutritional strategy can be conceptualized as a sequence of functional phases corresponding to predictable physiological changes across the day. Rather than delivering all nutrients together, this model organizes intake according to dominant metabolic states, allowing nutritional inputs to correspond with energy demand, substrate utilization, and recovery processes.
5.1 Activation Phase (Morning)
The transition from sleep to wakefulness is associated with increased cortisol secretion, sympathetic activity, and metabolic activation. During this period, mitochondrial pathways are oriented toward energy production to support physical activity, cognitive performance, and alertness following the overnight fast.
Nutritional inputs delivered during the morning phase may promote mitochondrial function, cognitive readiness, and circulatory efficiency while facilitating the transition from fasting physiology to active metabolism. This phase is primarily associated with energy mobilization and metabolic activation.
5.2 Regulation Phase (Daytime)
The daytime phase is characterized by active nutrient intake and metabolic processing. Insulin sensitivity is generally higher during this period, while digestive and absorptive functions are optimized for efficient utilization of dietary substrates.
The daytime metabolic profile is characterized by elevated activity in glycemic regulation and antioxidant pathways. Synchronizing nutrient delivery with this peak diurnal phase optimizes substrate utilization, maintains enteric integrity, and upholds physiological performance.
5.3 Recovery Phase (Night)
The night phase is associated with parasympathetic predominance and reduced energy expenditure. As melatonin levels rise and cortisol declines, cellular activity becomes increasingly oriented toward repair and restoration processes.
Functions including protein synthesis, membrane remodeling, and regenerative signaling become more prominent during this period. Nutritional inputs delivered during the recovery phase may contribute to neurotransmitter balance, cellular maintenance, and restoration of metabolic homeostasis following daytime metabolic demand.
This phased approach illustrates the temporal organization of metabolism relative to nutrient intake. By coupling delivery to circadian rhythms, the model synthesizes transport dynamics and fluctuating physiological requirements into a structured nutritional framework.
- From Ingredient-Based to System-Based Nutrition
The distinction between conventional supplementation strategies and circadian-aligned nutritional approaches reflects a broader evolution in how nutritional interventions are conceptualized. Traditional supplementation models typically rely on static dosing, where multiple nutrients are combined into a single formulation and administered without consideration of biological timing. Such approaches implicitly assume relatively stable metabolic responsiveness throughout the day.
In contrast, circadian-aligned strategies organize nutrient delivery according to predictable changes in metabolic state across the 24-hour cycle. Rather than emphasizing simultaneous administration of multiple compounds, this model considers how nutrient timing, transport characteristics, and functional roles interact with circadian physiology.
As illustrated in Figure 5, conventional approaches are generally formulation-centered and uniform in delivery, whereas circadian-aligned strategies are organized around temporal physiology and phase-specific metabolic demand. This perspective shifts emphasis from isolated biochemical effects toward coordinated nutritional organization across activation, metabolic processing, and recovery phases.
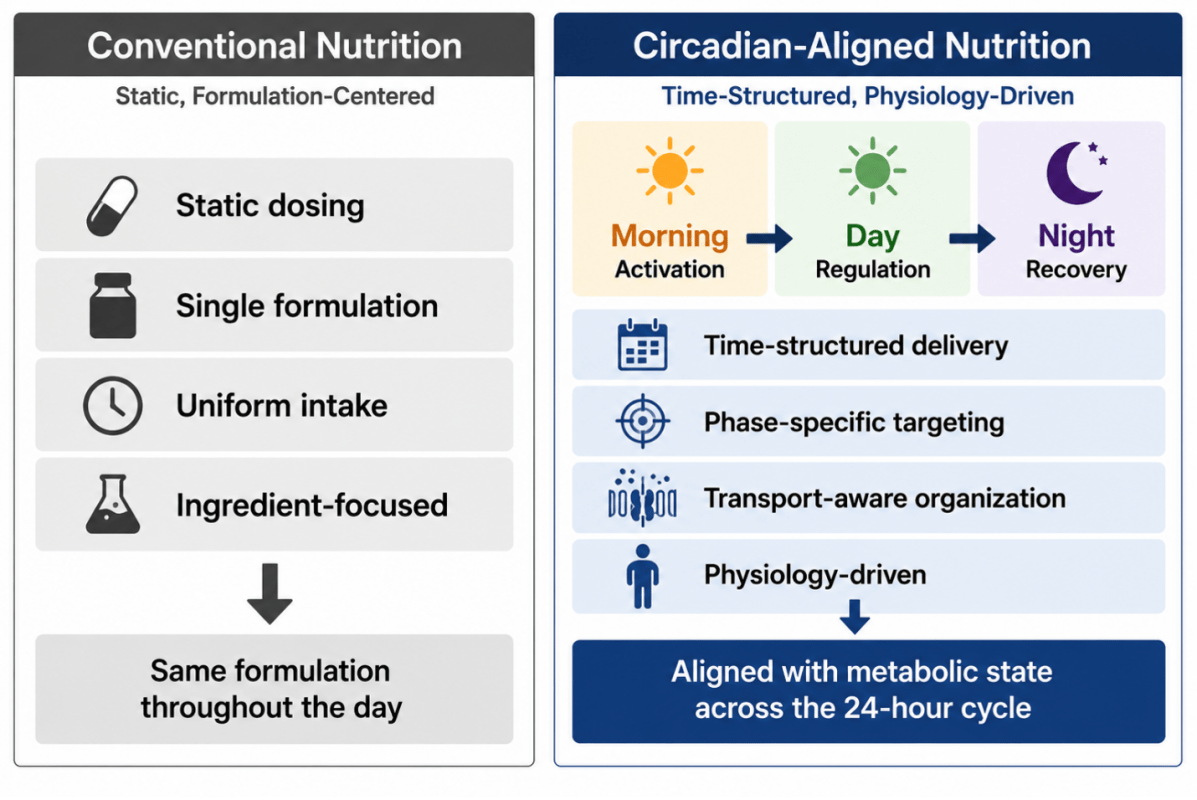
Figure 5. Conventional versus circadian-aligned nutritional organization.
Traditional supplementation strategies generally rely on static dosing and uniform nutrient delivery throughout the day. Circadian-aligned approaches organize nutrient intake according to temporal changes in metabolic state, integrating nutrient timing, transport characteristics, and phase-specific physiological demand.
Importantly, circadian alignment supplements rather than replaces established principles of nutrient composition and dosage. It introduces temporal organization as a critical variable modulating nutrient utilization and metabolic efficiency. Within this framework, nutrient timing, transport specificity, and delivery sequence function as integrated components of systemic metabolic regulation.
- Limitations and Future Directions
Although circadian biology provides a strong mechanistic rationale for temporally organized nutritional strategies, several limitations should be considered. First, substantial interindividual variability exists in circadian phase, commonly described as chronotype, which influences the timing of metabolic activity, hormonal rhythms, and sleep–wake behavior. Consequently, optimal timing of nutrient delivery may differ among individuals.
Second, environmental and behavioral factors including light exposure, sleep disruption, physical activity, and meal timing can significantly influence circadian alignment. These variables affect both central and peripheral clock systems and may alter metabolic responsiveness under real-world conditions, complicating the implementation of standardized approaches.
Third, despite a substantial mechanistic foundation, clinical evidence evaluating multi-phase nutritional systems remains limited. Most human studies have focused on meal timing, circadian misalignment, or isolated nutrients rather than integrated nutritional strategies organized across defined metabolic phases.
Future investigation should focus on translating circadian principles into clinically testable nutritional models. Areas of interest include controlled evaluation of phase-specific supplementation strategies, temporal organization of nutrient delivery, and the influence of nutrient form and transport pathways on metabolic outcomes across different circadian states. Additional clinical evidence will be necessary to determine the practical relevance and long-term applicability of system-based nutritional approaches.
- Conclusion
Circadian rhythms represent a fundamental organizing principle of human physiology, influencing metabolism, energy balance, hormonal regulation, and cellular repair across the 24-hour cycle. Aligning nutritional inputs with these biological rhythms provides a physiologically coherent approach that extends beyond traditional models focused primarily on nutrient composition.
A circadian perspective positions nutrition as a dynamic interaction with metabolic state rather than a static biochemical input. Within this framework, nutrient timing, delivery sequence, transport characteristics, and functional alignment become integrated variables that may influence utilization efficiency and metabolic responsiveness.
The transition from ingredient-centered supplementation toward time-based nutritional organization reflects a broader integration of temporal biology into metabolic science. As understanding of circadian physiology continues to evolve, time-structured nutritional strategies may provide a foundation for more coordinated approaches to metabolic and functional health.
References:
- Bass J, Takahashi JS. Circadian integration of metabolism and energetics. Science. 2010;330(6009):1349–1354.
- Daniel H. Molecular and integrative physiology of intestinal peptide transport. Annu Rev Physiol. 2004;66:361–384.
- Fernstrom JD. Large neutral amino acids: dietary effects on brain neurochemistry and function. Amino Acids. 2013;45(3):419–430.
- Morris CJ, Yang JN, Scheer FAJL. The impact of the circadian timing system on cardiovascular and metabolic function. Prog Brain Res. 2015;199:337–358.
- Panda S. Circadian physiology of metabolism. Science. 2016;354(6315):1008–1015.
- Pan X et al. Diurnal rhythm of H+/peptide cotransporter in rat small intestine. Am J Physiol Gastrointest Liver Physiol. 2002;283(1):G57–G64.
- Pan X et al. The diurnal rhythm of the intestinal transporters SGLT1 and PEPT1 is regulated by feeding conditions in rats. J Nutr. 2004;134(9):2211–2215.
- Peek CB et al. Circadian clock NAD+ cycle drives mitochondrial oxidative metabolism in mice. Science. 2013;342(6158):1243417.
- Scheer FAJL et al. Adverse metabolic and cardiovascular consequences of circadian misalignment. Proc Natl Acad Sci U S A. 2009;106(11):4453–4458.
- Takahashi JS. Transcriptional architecture of the mammalian circadian clock. Nat Rev Genet. 2017;18(3):164–179.
Tavakkolizadeh A et al. Diurnal rhythmicity in intestinal SGLT1 function, Vmax, and mRNA expression topography. Am J Physiol Gastrointest Liver Physiol. 2001;
