Aerobiotix, a rapidly growing global medical company has raised a $25 million of growth equity financing.
The round was led by Vivo Capital with participation by Asahi Kasei Ventures. The new funding will be used to accelerate sales, marketing, and new product development. The Company’s infection control products have been used to support thousands of procedures across multiple surgical specialty disciplines and in over 175 hospitals and multiple long-term care facilities.
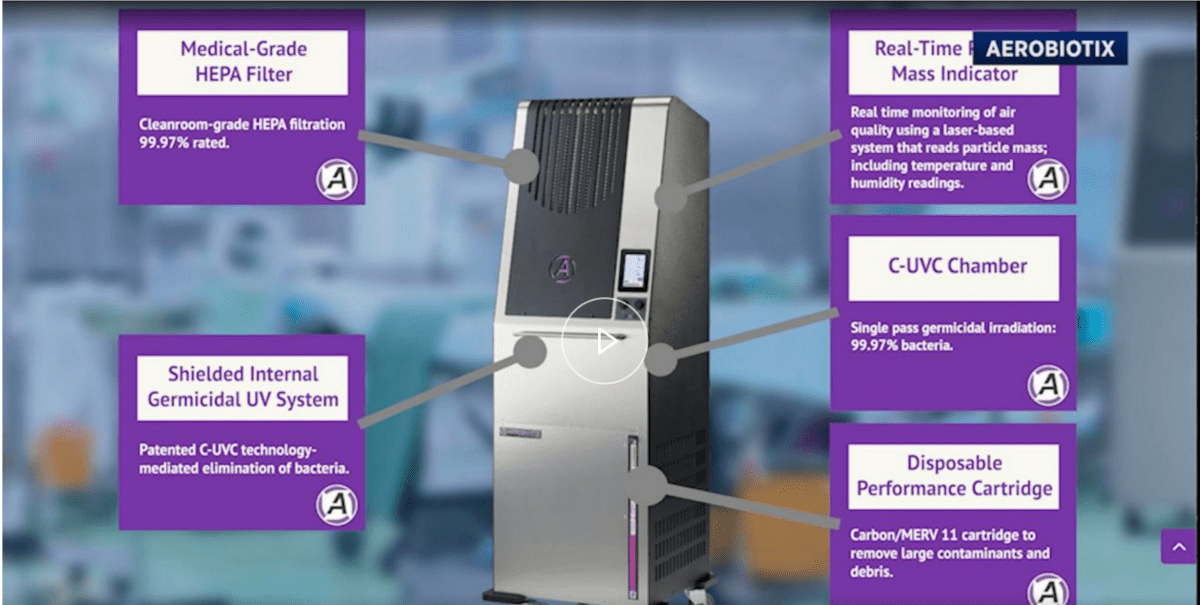
Aerobiotix has developed a novel suite of FDA 510(k) cleared hospital-based air decontamination products and its devices are supported by multiple peer-reviewed research studies. Aerobiotix devices, such as the operating-room based ILLUVIA® system, are used by leading healthcare centers to reduce bacterial and viral contamination, including SARS-CoV-2 and other emerging pathogens. Aerobiotix products provide mechanical and ultraviolet filtration to remove airborne threats with high efficiency to protect healthcare staff and visitors, and to reduce surgery-related infection risk for patients.
“Healthcare doesn’t stop at the patient, it encompasses an environment which is as important to outcomes as direct interventions. As we transition to a post-pandemic world, we are reminded of the long-term need for better environmental stewardship in healthcare and other critical settings. Aerobiotix is on the vanguard of this approach with direct decontamination, real-time air quality monitoring and internet of medical things connectivity,” states Dr. David Kirschman, founder and CEO of Aerobiotix.
Joseph Siletto, Managing Director at Vivo Capital noted, “Infection control has been an increasingly important area of focus for healthcare institutions. In particular, finding ways to reduce surgical site infections is a key priority since SSIs are extremely costly to patients, caregivers and healthcare systems. The Aerobiotix line of products provide healthcare institutions an effective, clinically proven way to improve safety and improve quality of care.”
“We have been very impressed by the Aerobiotix team and their ability to rapidly rollout a suite of innovative infection control products. The strong commercial traction Aerobiotix has achieved with limited outside capital attests to the value of the Aerobiotix solution. We are excited to partner with Aerobiotix to accelerate growth both domestically and internationally,” added Nathan Dau, Executive Director at Vivo Capital.