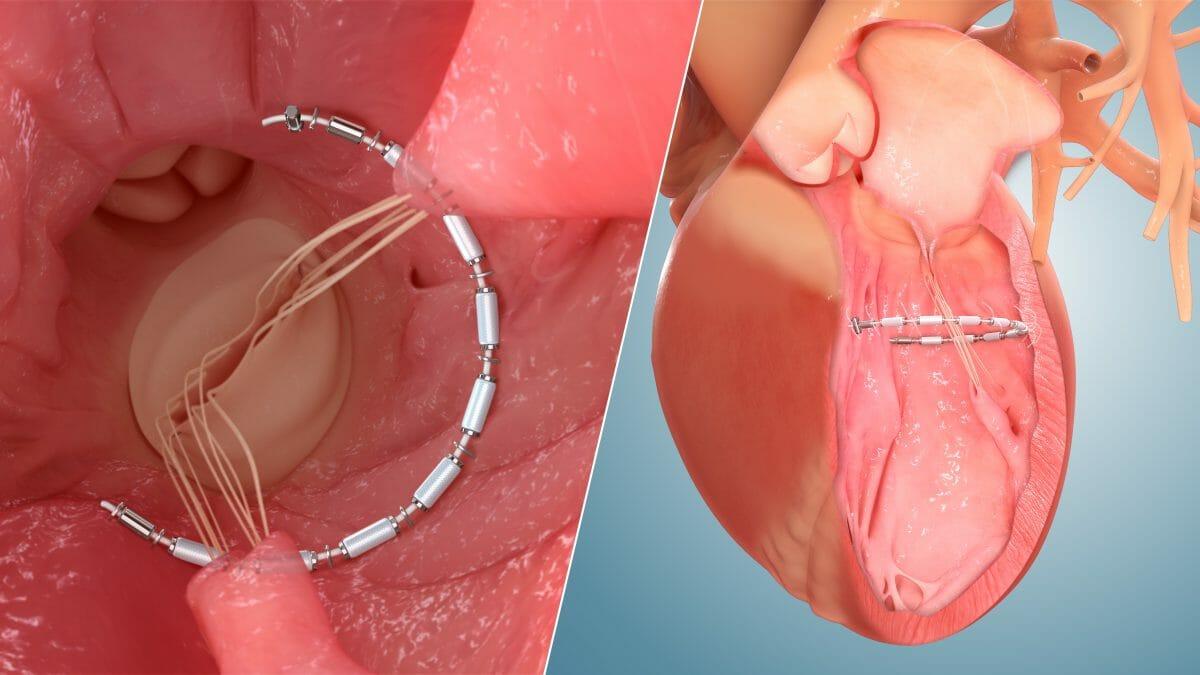
Ancora Heart, Inc., a medical device company developing a novel transcatheter device-based therapy to address heart failure (HF), and egnite, Inc., a leading digital health company in the field of cardiovascular care, today announced a strategic partnership to accelerate clinical trial enrollment for Ancora Heart’s CORCINCH-HF pivotal clinical trial, which is evaluating the safety and efficacy of the AccuCinch® Ventricular Restoration System. The partnership brings together the cutting-edge technology and expertise of both organizations with the goal of delivering innovative therapies to patients faster. The Christ Health Network in Cincinnati, OH is the first site in Ancora Heart’s CORCINCH-HF trial to utilize egnite’s Trial Accelerator artificial intelligence (AI) technology to help identify eligible patients.
“Our goal with the development of the AccuCinch System is to provide a minimally invasive treatment option to further improve the lives of heart failure patients whose symptoms progress despite guideline directed medical therapy,” said Mark Miles, chief commercial officer of Ancora Heart. “This partnership with egnite and The Christ Health Network will allow us to deploy AI tools such as egnite’s Trial Accelerator and expedite identification of eligible candidates for the CORCINCH-HF trial.”
egnite’s Trial Accelerator technology will also be leveraged at other participating CORCINCH-HF sites to drive patient identification and enrollment for the pivotal trial. The innovative solution can accelerate clinical trial enrollment through AI and big data processing, automatically screening hundreds of thousands of patients against trial parameters to help identify more trial candidates without hospital staff dedicating hours to manual chart review.
“Identifying patient candidates for clinical trial participation can be a complex task that requires many hours to review medical charts in the search for matching clinical criteria,” said Dr. Dean Kereiakes, president of The Christ Hospital Heart and Vascular Institute and co-principal investigator for CORCINCH-HF. “The use of AI technology streamlines the trial screening process by identifying candidates more quickly and efficiently, thereby freeing up critical resources needed for clinical trial success.”
“We are thrilled to partner with leading life sciences organizations like Ancora Heart and apply our novel technology to accelerate clinical trial enrollment,” said Joel Portice, president and chief executive officer of egnite. “We believe this collaboration is a significant step forward for patients and are excited to be at the forefront of cardiovascular care.”