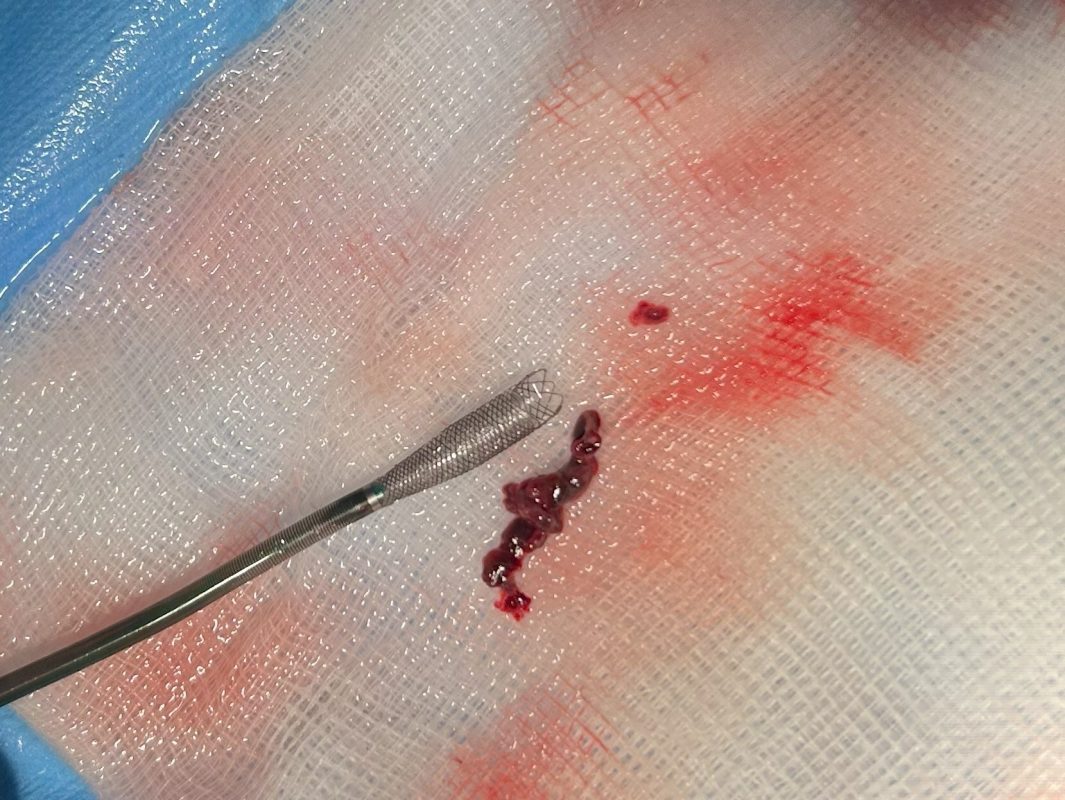
Ceretrieve announced today the successful results of the company’s multicenter, single-arm study, showcasing the capabilities of its state-of-the-art aspiration catheter. The study, conducted across two centers, included 20 patients who suffered from acute ischemic stroke (AIS) due to intracranial large vessel occlusion (LVO) and were eligible for thrombectomy within 24 hours of symptom onset. The study was aimed for assessing the safety and initial performance of the Ceretrieve Device.
Redefining Stroke Treatment
Ceretrieve’s mission is to transform stroke care, save lives, minimize disability, and significantly improve post-stroke quality of life. Ceretrieve’s aspiration catheter is at the forefront of this mission, delivering aspiration capabilities that are far superior when compared to existing devices, allowing clot(s) removal in a single pass, while fully restoring blood flow. Designed for seamless integration within a conventional 6Fr delivery catheter, Ceretrieve’s device offers exceptional trackability and maneuverability, ensuring efficient access to the clot location.
As shown in the study conducted, Ceretrieve’s aspiration catheter not only excelled in performance but also ensured the highest safety standards by reducing the risk of releasing clot fragments further into the brain. In the first-in-human (FIH) study 80% Complete/Near-Complete Perfusion was achieved.
The study included two generations of the device, and the results with the improved Gen 2 device are highly impressive: 100% Complete/Near-Complete Perfusion achieved in all treated patients; and 83% First Pass Complete Perfusion (FPE mTICI 3), far exceeding the 30%-40% rate of current gold-standard devices.
Maysa Mustafa, CEO of Ceretrieve:
“The promising results of our clinical studies are the culmination of years of hard work and R&D. Our mission to redefine stroke treatment drives everything we do, and these results signify a major leap forward in saving and dramatically improving lives following an ischemic stroke.”
“The ‘first pass effect’ has been shown to improve patient outcomes” says Dr. Shady Jashan (Galilee Medical Center, Israel) who performed the first neuro thrombectomy procedure with the Ceretrieve device. “Our experience in FIH trials demonstrated that the Ceretrieve device quickly enables complete clot ingestion in first pass even for the most challenging clots and anatomies. This has the potential to dramatically improve patient clinical outcomes.”
Prof. Serder Geyik, MD (Florya Medical Park, Turkey): “The term ‘Everything comes with a price’ is not valid for the Ceretrieve device. Ceretrieve’s device aspirates the clot with a giant bore tip- double than the largest catheters in the market, for maximal vacuum effect without paying the price in trackability. In addition, it provides local flow restriction in the middle cerebral artery reducing distal emboli. It is the only device that includes all the features for successful thrombectomy and first pass effect in one single device. ”
Amir Belson, M.D., Ceretrieve Chair:
“With Ceretrieve’s advanced technology, we are seeing significant improvements in patient recovery rates. This innovation is setting a new standard in stroke care, offering patients better outcomes and real hope for recovery.”