Moby Cutter™

Announcing the second-year creation, of the amazing Moby Cutter™. It’s one of the safest surgical wire cutters on the market today!
The Moby is produced by Stanley Medical Designs, Inc., a company dedicated to making surgical procedures safer, more efficient, to improve the patient care experience. The Moby, is patented as a new hybrid surgical wire cutter, with a catchment device. The catchment device is an optional, transparent basket of bullet-resistant material class, called a Flick Catcher™. Like a catcher’s glove, “it catches and retains cut pieces™”. This optional Flick Catcher™, can prevent cut pieces of material from becoming dangerous projectiles, which can puncture skin, endanger eyes or fall into the deep surgical field, preventing a sentential event.
Our medical design team has over 35 years of experience performing surgical procedures in Hospitals, Emergency Rooms and Urgent Care Systems. It has been cleared for commercialization per FDA guidelines and sold as a prescription only (Rx) medical device.
The Moby Cutter™ Features
- 2 mm standard wire, cutting capacity, measuring device included
- 6 ½ inches tall
- Ergonomic handles for improved grip
- Dual action return spring to prevent hand fatigue
- Beveled blade edges allow closer cutting to the skin surface, avoiding tissue damage
- Safety lock
- Surgical Steel composition
- Weight 0.25 Kg
- Optional single-use basket attachment, helps to prevent a Sentinel Event
- Totally autoclavable stylish device
- FDA Registered and listed as a class 1 medical device
- Available in a Surgical Green color
The Moby Cutter™ Has Applications In
- Orthopedic Surgery (Cutting K- wires and select Steinmann Pins)
- Hand Surgery (Cutting K- wires)
- Podiatry Surgery (Cutting K- wires)
- Foot and ankle Surgery (Cutting K- wires)
- Cardiothoracic Surgery (Cutting Sternal wires)
- Oral Facial Maxillary Surgery (Cutting jaw wires)
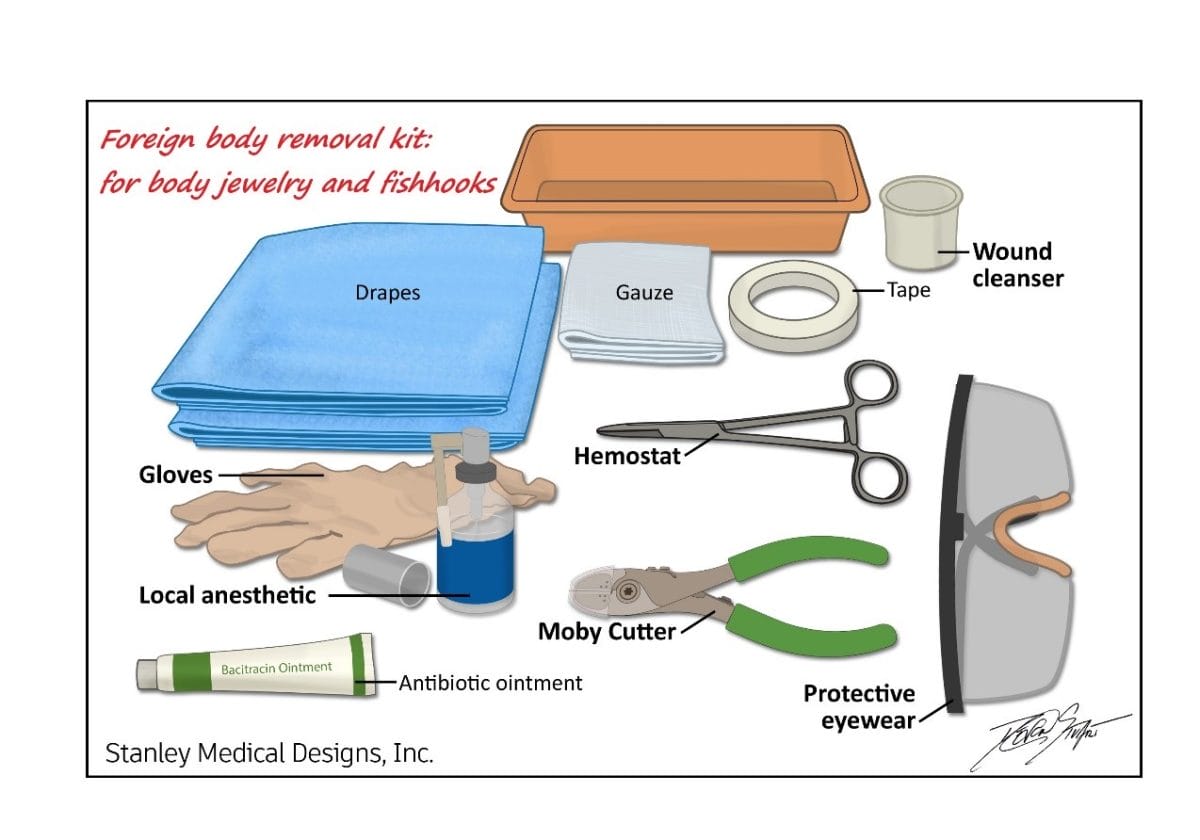
- Emergency Room (foreign body removal, cutting body jewelry and fishhooks)
- Urgent Care (foreign body removal, cutting body jewelry and fishhooks)
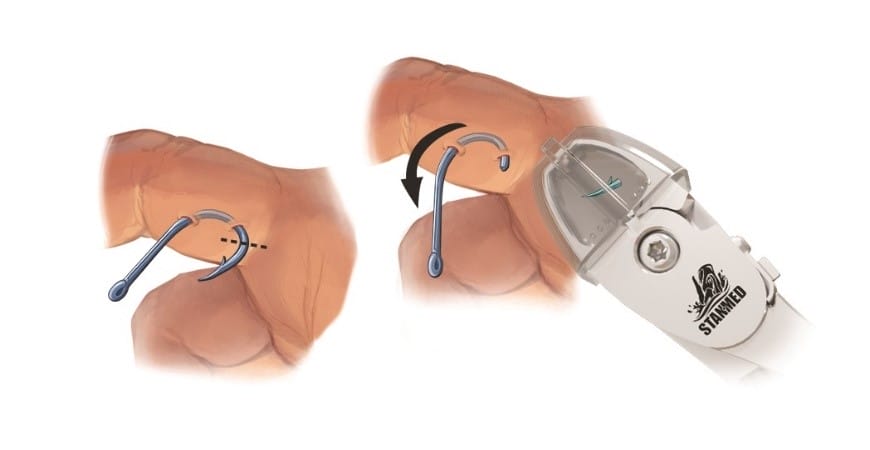
“The Moby Cutter™ has special recognition from the US Patent office, for usage in surgical fishhook removal procedures, which distinguishes it from the unsterile standard hard ware store wire cutters frequently being used.”
The Moby Cutter™ has a sister cutter, Called the SoBe Cutter™, named in tribute to our company’s South Beach, Florida origin. Both cutters have similar features and function. The limited edition, SoBe Cutters™ comes in stunning Vice Blue or Vice Red colors.
Every surgeon must have one on their surgical tray. Every Urgent Care facility should have one in their procedure supply room. Be one of the first to discover a new cutting experience!
More product information can be found here or email. View of YouTube.
Editor’s Note: Anthony G. Stanley, MD, Bio: Staff physician CritiCare Clinics (Urgent Care) Miami, Fl. Staff Emergency Room Physician, Baptist Healthcare of South Florida. Medical Device Inventor (holder 6 medical device patents), Expert Medical Witness, and Author.
Dr. Stanley received his medical degree from Rutgers-New Jersey Medical School, Newark New Jersey. He completed Residency in Internal Medicine at University of Miami-Jackson Memorial Medical Center/ Miami VA Medical Center Miami, Florida.

