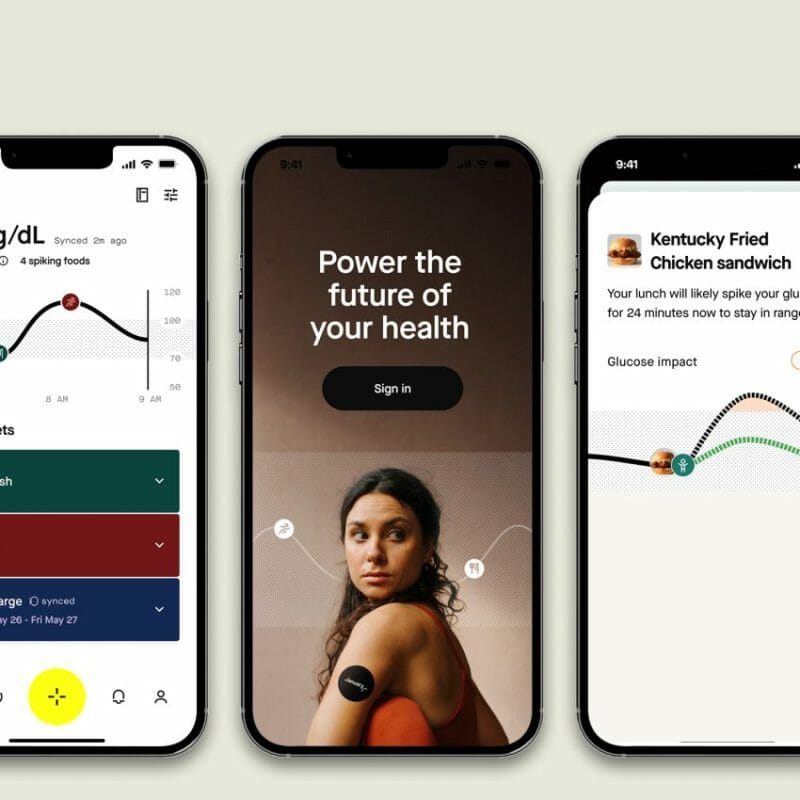
Today, precision health tech company, January AI, releases a new generative AI-enabled app that estimates and predicts individuals’ glucose response to 32 million foods, even when members are not wearing a continuous glucose monitor (CGM). January AI can help anyone who wants to discover the intricacies of their body and use data to support a healthier lifestyle. And beyond glucose prediction, January AI shifts key behaviors such as intermittent fasting, increasing fiber consumption, aligning calorie intake with activity level, getting post-meal movement, and reducing glucose-spiking foods. January AI is available now at app.january.ai for $288 for the first month ($28 monthly membership fee after).
Unlike a normal CGM, January AI predicts how food will impact your blood sugar, beyond only reporting what’s happened. “Think of it as a weather report. If it only told you it’s already raining, you wouldn’t find it very useful,” said Noosheen Hashemi, who co-founded January AI in 2016 with Stanford University’s Director of Genomics, Dr. Michael Snyder, PhD. “With January AI, members enter foods they are considering eating, and the predictive AI model will tell them what it will do to their blood sugar. It helps them make better decisions about what they’re about to put into their body, kind of like having an AI nutritionist in your pocket to let you know whether you should order a smoothie or an arugula salad, and what happens if you take a 25-minute walk afterwards.”
Metabolic Health, Meet Artificial Intelligence
The launch of their new app marks January AI as the first and only metabolic health company to harness the power of artificial intelligence to precisely predict individuals’ glucose response. Their application of AI advances the growing consumer CGM category, driven by research which shows that managing blood sugar is key to good health, even for people without diabetes, due to glucose level’s correlation to weight regulation, cholesterol, inflammation and more.
“The metabolic crisis is far more prevalent as an endemic than COVID was as a pandemic,” Dr. Michael Snyder said in a recent interview. “Getting glucose under control is a big, big deal. Nearly 100 million people are walking around with prediabetes, and 90% don’t even know it. If we don’t measure people’s health, does it mean they’re healthy?”
January AI is democratizing access to the powerful intelligence provided by continuous glucose monitors, that few can afford today, by deploying AI to make CGM use optional after 14 days, which reduces costs and discomfort from constant CGM use.
January’s AI and 32 million database of foods allows members to see their precise predicted glucose curve projected up to two hours into the future, the world’s first innovation that increases glucose prediction from one to two hours, and makes January AI the first to offer predictive capabilities to consumers without diabetes.
Health is the Product of Millions of Data Points
January’s AI uses a continuous glucose monitor (CGM) and activity tracker to provide unprecedented visibility into the body, empowering members to make the best choices for their health. It integrates data from partners including Oura Ring, Apple Health and Fitbit to further analyze members’ precise health data and provides insights and recommendations to reach their goals whether that is to lose weight or improve their total metabolic health.
January AI aids with calorie alignment to activity by using data to dynamically figure how many calories a member should consume. It tracks post-meal activity to encourage members to move immediately after eating, a habit shown in their research to lower blood sugar. Fasting is another key lever for improving insulin sensitivity and January AI frictionlessly tracks fasting periods and slowly nudges members towards appropriate fasting periods.
By analyzing lifestyle choices, such as how close to bedtime you ate, January supports and promotes better sleep, which is foundational to cardiometabolic health. “We are excited to work with companies like January AI to provide sleep insights to their members,” said Tom Hale, ŌURA CEO. “January’s last-meal-sleep-gap helps members gain awareness about how their mealtime choices can affect the quality of their sleep,” he continued.
January can give you macros, track your fiber intake, and keep you accountable and motivated through gentle nudges and daily reminders that support your goals so you stay on track. The best part is that January makes it easy by offering nutritional values for local restaurants and leveraging voice tracking capabilities – simply tell January what you ate and it does the rest.
How it Works
Get the hardware: Complete a short telehealth evaluation to get your continuous glucose monitor (CGM) prescription. Once approved, January will send your CGM to your door.
Train the AI: Download the January AI app. After one week, January’s AI has enough data to establish a baseline of your health and model your physiology. January AI’s proprietary artificial intelligence algorithms have been trained on the reactions of different individuals to 32 million different foods. January AI’s new app will predict the way that an individual will respond to most other foods.
Receive insights: Start getting glucose predictions and real-time insights to align key behaviors around your metabolic health. This intelligence helps minimize sugar spikes in the bloodstream by substituting some foods, or by first eating other foods that interact with one’s individual metabolism in ways that are likely to help suppress those spikes, reducing the effects of foods likely to cause big spikes.
CGM optional: After 14 days, January’s AI no longer requires a CGM to display your glucose curve, insights, and predictions.