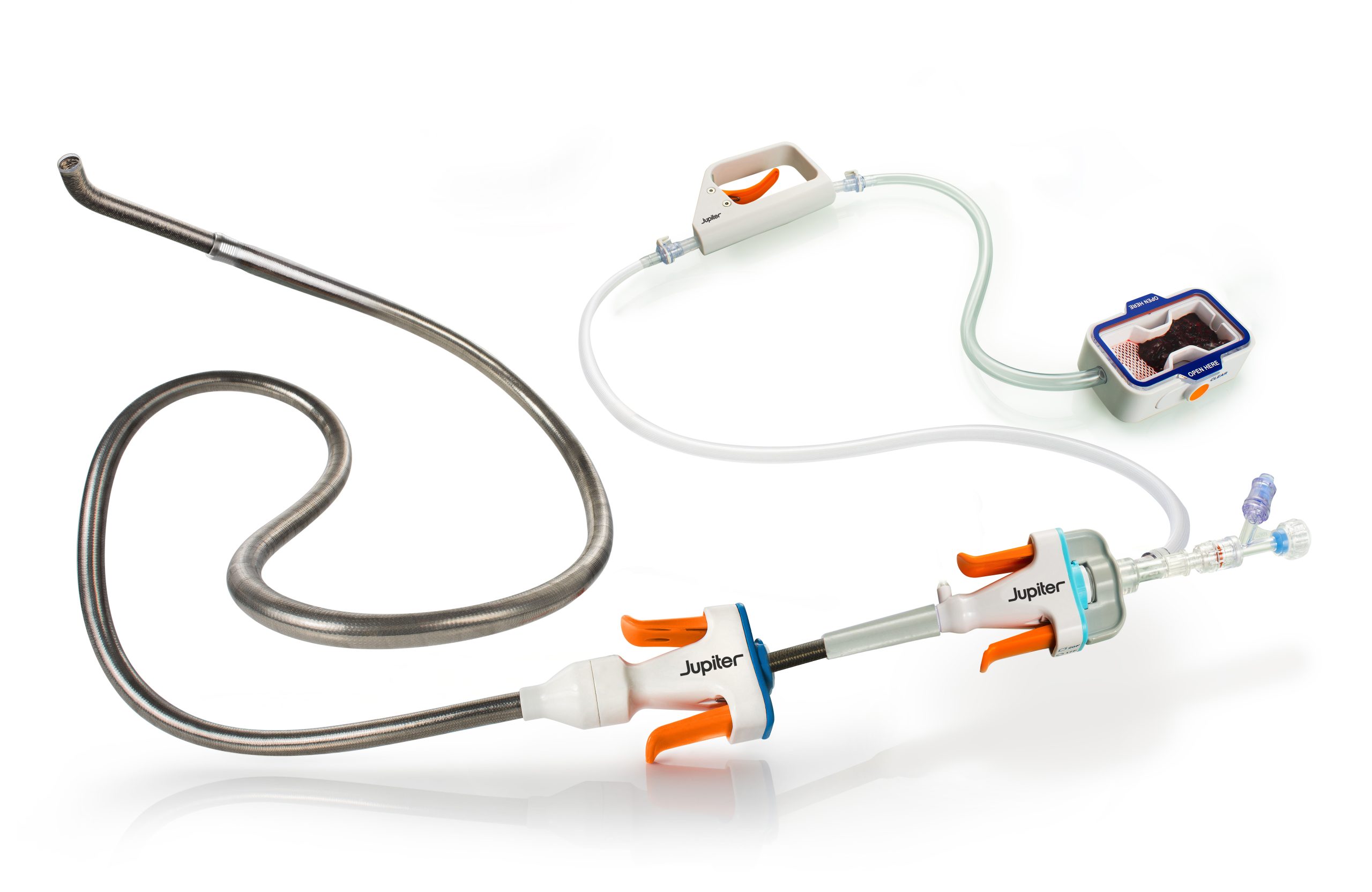
Vertex Pulmonary Embolectomy System demonstrated significant right heart recovery, low adverse event rates, and reproducible performance across high-severity PE patients
TIME Names Xenco Medical one of the TIME100 Most Influential Companies in the World and the Winner of the 2026 TIME100 Impact Award in Health
Xenco Medical was honored by Time as the sole recipient of the TIME100 Impact Award in Health in 2026, signifying its leading, global distinction in impact on healthcare