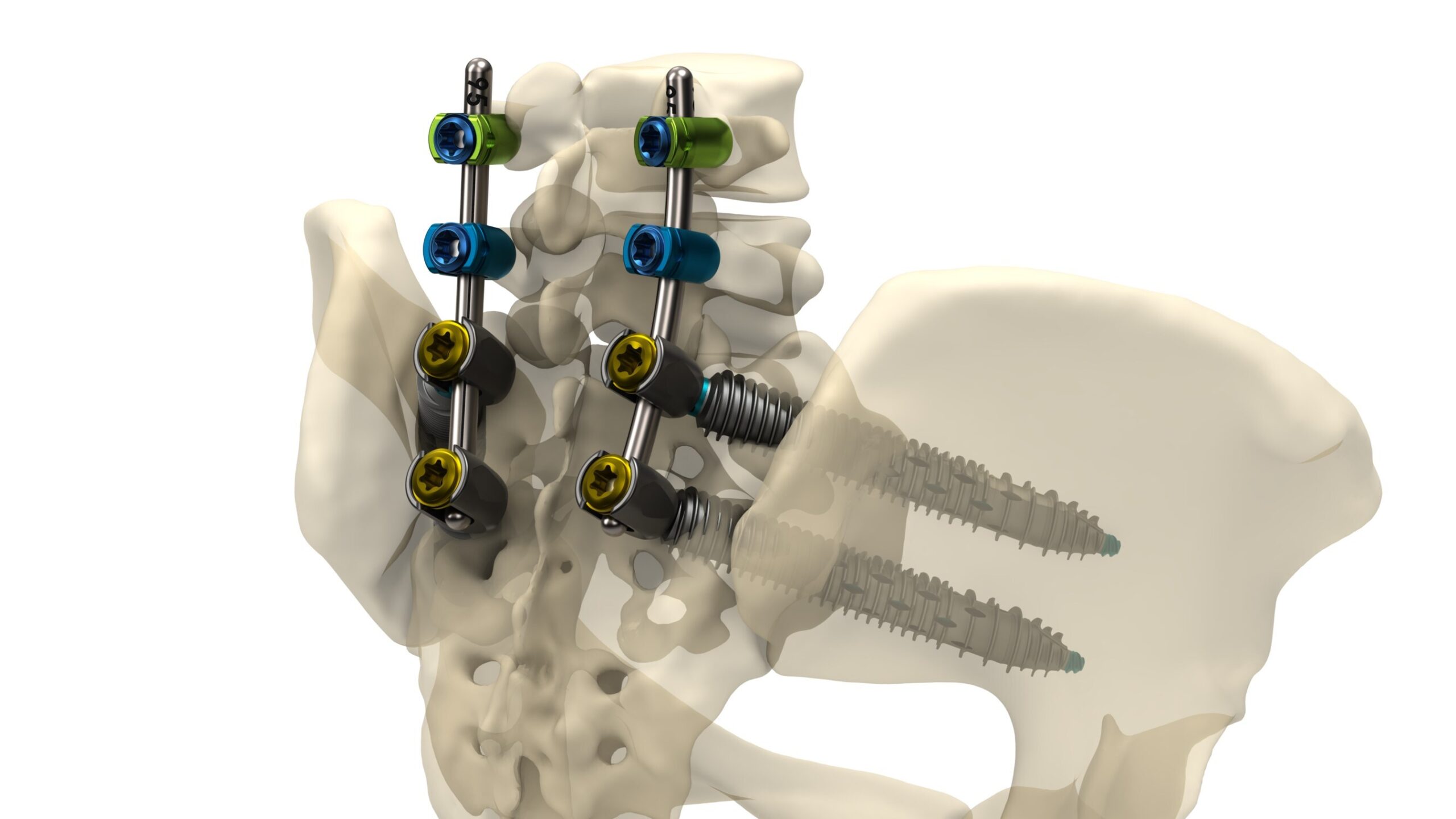
Inaugural case performed by spine surgeon Dr. Scott L. Parker highlights the system's strength, versatility, and precision for supporting multi-level spinal constructs
Medical Device News Magazine is committed to delivering high‑quality, in‑depth medical device news and biotechnology updates that include expert bylines. Our coverage offers a comprehensive view of the trends, innovations, and breakthroughs shaping the future of medical technology.
We also strive to provide a distinctive perspective on the rapidly evolving healthcare landscape. By staying connected with our reporting, you gain access to exclusive insights on industry developments, emerging technologies, and global regulatory changes that influence the medical device sector. At the same time, you join a vibrant community of professionals, innovators, and thought leaders dedicated to advancing healthcare through cutting‑edge solutions.
We invite you to explore our wide range of content—from the latest product announcements to detailed analyses of industry trends. And if our mission aligns with your goals, we offer opportunities for advertising and collaboration, helping you connect with a highly engaged audience of medical technology professionals.
Definition medical devices.
Medical Device Industry News | Latest Developments, Breakthroughs & Future Outlook
Latest News and Developments
Cartessa Aesthetics and Classys Introduce QUADESSY, a Faster, Safer and More Versatile Microneedling Radiofrequency Platform
Quadessy delivers multi-layer skin remodeling through a unique tip and dual-depth design
Medtronic and Merit Medical Systems enter into agreement to offer new, FDA-cleared ViaVerte™ system, a basivertebral nerve ablation treatment for chronic vertebrogenic lower-back pain
Medtronic's latest partnership with Merit Medical Systems introduces the groundbreaking ViaVerte™ BVNA system—an innovative solution for chronic vertebrogenic pain
Clinical Trials
Complement Therapeutics Announces First Patient Dosed with CTx001 in the Phase I/II Opti-GAIN Study for Geographic Atrophy Secondary to AMD
First patient dosed in Opti-GAIN, a first-in-human Phase I/II clinical trial of CTx001 in Geographic Atrophy secondary to AMD
Innovative Medical Equipment, LLC Highlights Published VA Clinical Study on Thermal Therapy and Pain Treatment Utilization
The study, published in The March 2026 edition of Federal Practitioner, reported an association between thermal therapy use and reduced opioid and benzodiazepine utilization, as well as lower healthcare utilization in the patient population.
Results from PARADIGM Phase 2b Trial of PrimeC in ALS Demonstrating Meaningful Clinical Outcomes and Biological Activity Published in JAMA Neurology
The Journal of the American Medical Association (JAMA Neurology) publication highlights consistent clinical and biomarker findings from NeuroSense PARADIGM Phase 2b trial in ALS, including slower functional decline, reduced risk of ALS-related complications, and modulation of disease-relevant biomarkers
Biotechnology News
iXCells Biotechnologies Joins International Collaboration to Evaluate Chemotherapeutic Toxicity in Human Organoids
Joint initiative with Rosebud Biosciences, Kantify, and funded by Incite will identify and quantify off-target toxicities in chemotherapy patients
Primerdesign Partners with Origin Sciences to Create Novel Rectal Mucosal Extraction Workflow
Robust DNA extraction supports earlier detection and diagnosis of colorectal cancer and other gastrointestinal diseases
AnalytiChem Launches New Legionella Testing Selective Agar Plate
Redipor® Legionella agar plate range - specifically designed to support the detection and enumeration of Legionella in water samples
Mergers & Acquisitions
Funding
Gestalt Diagnostics Raises $7.5 Million Series A Funding | to Expand AI-Powered Pathology Platform
The round was led by Cowles Ventures, TVF Funds, Inland Imaging Investments, KickStart Funds, and prominent angel investors from the Pacific Northwest.
Intensity Therapeutics, Inc. Announces $2.35 Million Public Offering
The Series B-1 warrants will have an exercise price of $0.85 per share, will be immediately exercisable and will expire 5 years from issuance. The Series B-2 warrants will have an exercise price of $0.85 per share, will be immediately exercisable and will expire 18 months from issuance.
Industry Expert Bylines

Plaque Quantification In Coronary Arteries Is a ‘Game-changer’ | By Campbell Rogers, M.D., F.A.C.C., Executive Vice President & Chief Medical Officer, HeartFlow
Artificial intelligence provides a simpler and faster alternative and could be a potential game-changer for their heart patients. For example, HeartFlow’s AI-enabled Plaque Analysis tool leverages an automated, deep learning-based, non-invasive method for identifying, characterizing, and segmenting plaque throughout the coronary arteries. By offering a more comprehensive view, plaque analysis tools can improve diagnostic accuracy, enhance treatment planning, and help stratify risk.

The Crossroads of a Revolution: Medical Device Design Meets Disruption | By Steve Levine, Senior Director, Virtual Human Modeling, Dassault Systèmes
Explore how the intersection of medical device design and disruptive technologies is reshaping the healthcare industry. Discover how new players armed with AI-driven devices and advanced manufacturing tools are pushing boundaries and revolutionizing the future of medical innovation. Steve Levine, Senior Director at Dassault Systèmes, shares insights on the next plateau of medical device design.

How Technology Is Driving Person-Enabled Brain Health, Fueling Better Outcomes and Options | By Mark Lehmkuhle, PhD, CEO, Founder of Epitel
In brain health, traditional inpatient monitoring systems present a myriad of barriers. They often require travel to a specialized health center, are cumbersome and restrictive for patients, and technical requirements and limitations impede providers’ ability to even provide such services. Today, advancements are disrupting this model to fuel the growth of person-enabled health and the ongoing treatment of chronic neurological disorders and conditions
FDA Medical Device Updates
Market Reports
Nanox Receives FDA Clearance for General Use of New Imaging System, Nanox.ARC X
First FDA clearance for Nanox.ARC X to produce tomographic images for general use, including musculoskeletal, pulmonary, intra-abdominal and paranasal indications. The enhanced imaging system features sleek design with smaller footprint; simplified ‘plug and play’ installation process and the system design enables software upgrades and new capabilities to be added remotely following future regulatory clearances
Medline Announces FDA Clearance for REFLEX HYBRID Nitinol Implants for Foot and Ankle Surgery
“REFLEX HYBRID further demonstrates our commitment to offering innovative solutions for foot and ankle surgeons. The first to market product addresses gaps in the current competitive landscape, including offering indication specific designs, intraoperative compression, and intraoperative adjustment with a nitinol implant,” said Scott Goldstein, vice president of product management for Medline UNITE.
Executives

Rad AI Names Demetri Giannikopoulos as Chief Growth Officer to Accelerate AI-Driven Radiology Healthcare Growth
Mr. Demetri brings over 20 years of experience in healthcare technology, with a track record of advancing AI adoption in complex clinical settings.
RapidRatings Appoints Charlie Minutella as CEO to Accelerate Strategic Growth and Innovation
Charlie Minutella brings a wealth of industry knowledge and a strong track record of leadership, innovation, and operational excellence. His appointment ushers in a new chapter for RapidRatings as it continues to shape the future of financial risk intelligence.

Dr Ray Kotwicki, Chief Medical Officer of Hightop Health, Appointed President of the Southern Psychiatric Association
Dr Ray Kotwicki will lead the SPA into its next chapter, focusing on education, collaboration, and advocacy for psychiatrists across the region.
Advancements In Imaging
KA Imaging Reports Record Revenue Plus Strategic Hires
KA Imaging reported a collection of positive metrics to its stakeholders, with revenue four times larger than that of the previous year and a 38% growth in headcount in 2023. The positive trend continues to be observed in Q1 2024.

Sectra Reports Enterprise Imaging Solution Now Covers Majority of Midwest Patients Following New Contract
Isaac Zaworski, President of Sectra advises: “Collaborating across sites and providing efficient radiology workflows are needed to cope with the increasing imaging volumes. This new customer is the 60th healthcare provider to use Sectra in the US Midwest and I’m pleased to be continuing our work in this area. With our local experience as well as our knowledge in providing multi-site solutions for large organizations we will help them on this journey in the best way possible.”
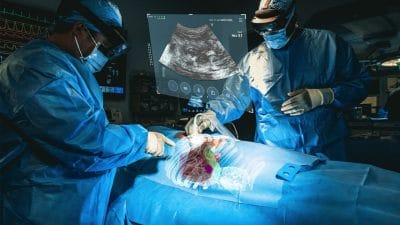
MediView Announces Study Results Demonstrating High Registration and Targeting Accuracy of Augmented Reality-Based Navigational Guidance System
MediView notes the system, which has received 510(k) clearance from the U.S. Food and Drug Administration and is currently in clinical use, is intended to be used adjunctively for minimally invasive ultrasound guided needle-based procedures for soft tissue and bone.
Hospitals In the News
Non-Profit News
Healthcare: Understanding the Business Side of the Industry and Its Implications

Top 10 Must-Have EMS Vehicle Equipment for Emergency Responders
Modern EMS vehicles require comprehensive medical equipment and supplies, with costs averaging around $19,000 per vehicle to ensure readiness for any emergency scenario.

How to Ensure Your Business Meets FDA Compliance Standards
Business Meets FDA Compliance Standards:

MHPAEA Act & NQTL Analysis And Rules
The U.S. Departments of Treasury, Labor, and Health and Human Services have recently released final regulations regarding nonquantitative treatment limitations (NQTL) under the Mental Health Parity and Addiction Equity Act (MHPAEA Act).
Vesilut Peptide: Potential Implications in Biological Systems and Scientific Research
Vesilut peptide may hold the key to advancing our understanding of physiological processes and developing innovative approaches to challenges in molecular biology and beyond.