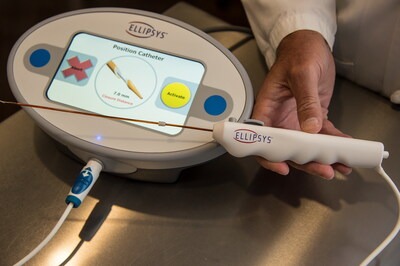
Ellipsys Medical, Inc. today announced that the Ellipsys® Vascular Access System will once again be commercially available, with initial availability anticipated in mid-2026. The announcement marks the planned reintroduction of the minimally invasive technology to clinicians and patients following a period of limited market availability. The Ellipsys System, previously available...
Medical Device News Magazine is committed to delivering high‑quality, in‑depth medical device news and biotechnology updates that include expert bylines. Our coverage offers a comprehensive view of the trends, innovations, and breakthroughs shaping the future of medical technology.
We also strive to provide a distinctive perspective on the rapidly evolving healthcare landscape. By staying connected with our reporting, you gain access to exclusive insights on industry developments, emerging technologies, and global regulatory changes that influence the medical device sector. At the same time, you join a vibrant community of professionals, innovators, and thought leaders dedicated to advancing healthcare through cutting‑edge solutions.
We invite you to explore our wide range of content—from the latest product announcements to detailed analyses of industry trends. And if our mission aligns with your goals, we offer opportunities for advertising and collaboration, helping you connect with a highly engaged audience of medical technology professionals.
Definition medical devices.
Medical Device Industry News | Latest Developments, Breakthroughs & Future Outlook
Latest News and Developments
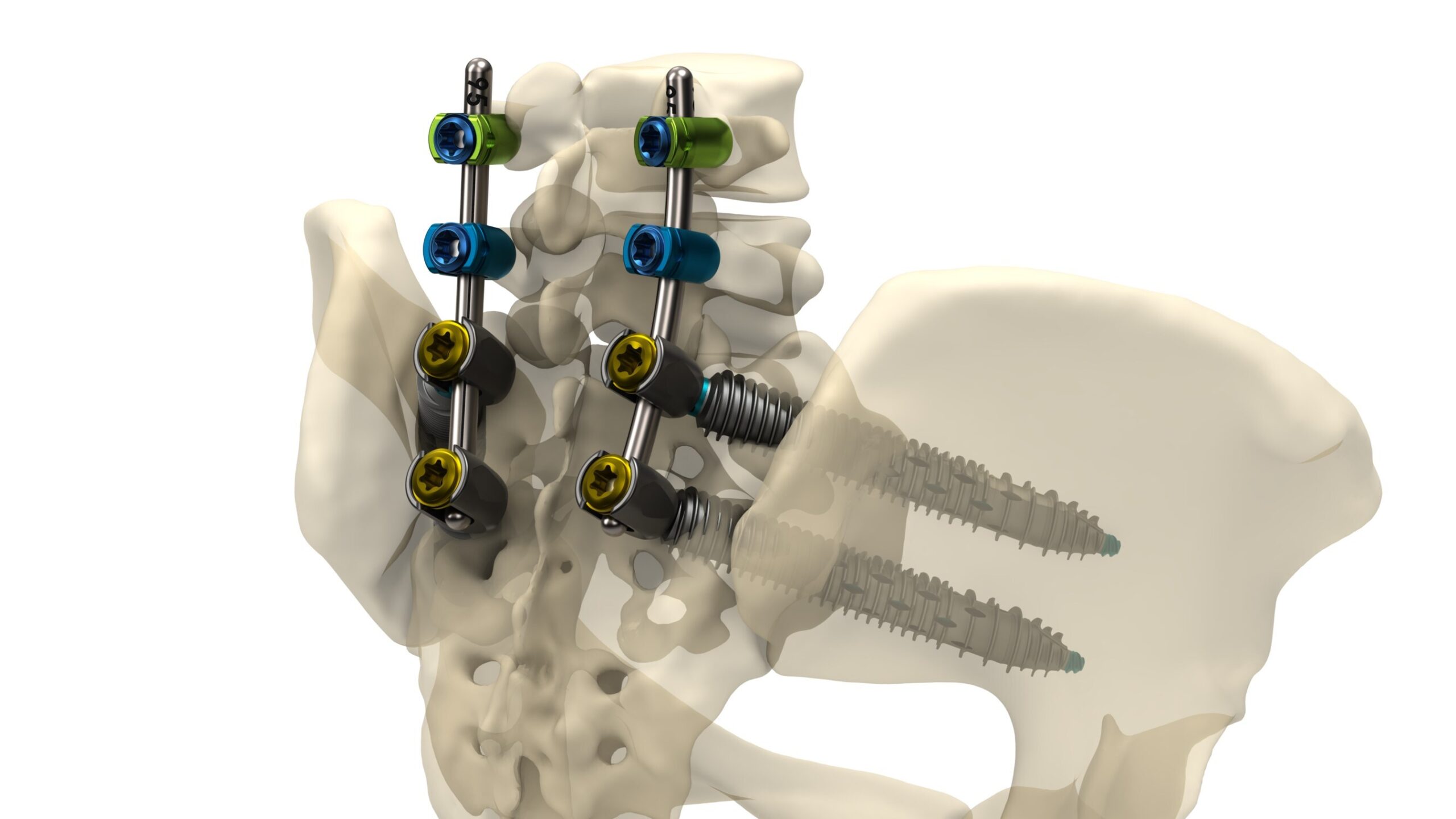
Genesys Spine Announces Launch of the SIros-X™ System for Sacroiliac Joint Fusion and Pelvic Fixation of Pedicle Screw Constructs
Inaugural case performed by spine surgeon Dr. Scott L. Parker highlights the system's strength, versatility, and precision for supporting multi-level spinal constructs
Cartessa Aesthetics and Classys Introduce QUADESSY, a Faster, Safer and More Versatile Microneedling Radiofrequency Platform
Quadessy delivers multi-layer skin remodeling through a unique tip and dual-depth design
Clinical Trials
Complement Therapeutics Announces First Patient Dosed with CTx001 in the Phase I/II Opti-GAIN Study for Geographic Atrophy Secondary to AMD
First patient dosed in Opti-GAIN, a first-in-human Phase I/II clinical trial of CTx001 in Geographic Atrophy secondary to AMD
Innovative Medical Equipment, LLC Highlights Published VA Clinical Study on Thermal Therapy and Pain Treatment Utilization
The study, published in The March 2026 edition of Federal Practitioner, reported an association between thermal therapy use and reduced opioid and benzodiazepine utilization, as well as lower healthcare utilization in the patient population.
Results from PARADIGM Phase 2b Trial of PrimeC in ALS Demonstrating Meaningful Clinical Outcomes and Biological Activity Published in JAMA Neurology
The Journal of the American Medical Association (JAMA Neurology) publication highlights consistent clinical and biomarker findings from NeuroSense PARADIGM Phase 2b trial in ALS, including slower functional decline, reduced risk of ALS-related complications, and modulation of disease-relevant biomarkers
Biotechnology News
iXCells Biotechnologies Joins International Collaboration to Evaluate Chemotherapeutic Toxicity in Human Organoids
Joint initiative with Rosebud Biosciences, Kantify, and funded by Incite will identify and quantify off-target toxicities in chemotherapy patients
Primerdesign Partners with Origin Sciences to Create Novel Rectal Mucosal Extraction Workflow
Robust DNA extraction supports earlier detection and diagnosis of colorectal cancer and other gastrointestinal diseases
AnalytiChem Launches New Legionella Testing Selective Agar Plate
Redipor® Legionella agar plate range - specifically designed to support the detection and enumeration of Legionella in water samples
Mergers & Acquisitions
Funding
Exero Medical Closes on $12.7M Preferred Seed Financing
This financing includes investments from new investors Longevity Venture Partners, Tech Council Ventures, and Edge Medical Ventures, alongside participation from existing investors notes Exero Medical.
REEV Raises $9.2M | To Revolutionize Mobility Assistance for Patients with Gait Disorders
“With the support of our world-class partners, we are thrilled to enter the next phase of clinical and industrial development,” said Amaury Ciurana, Co-Founder and CEO of REEV. “Our innovative technologies, like the DREEVEN motorized orthosis, aim to redefine mobility assistance, transforming lives and reshaping the orthotics industry.”
Industry Expert Bylines

How A Streamlined Quality Management System Can Transform the Prescription Ordering Journey for Patients | By Chuck Serrin, VP of MedTech and Life Sciences Industry Marketing at Propel
Chuck Serrin advises since the pandemic, there has been a rise in ordering everything to the home – from vitamins to prescription drugs – and the days of venturing to a brick-and-mortar store are declining. The pharmaceutical industry is no different. It too is evolving, and pharmacy-focused businesses must make sure their systems are updated to keep up with the changing times.

Bringing Innovation to Life in the Medical Technology Industry | By Shawn Luke, Technical Marketing Engineer, DigiKey
Shawn Luke is a technical marketing engineer at DigiKey. DigiKey is recognized as the global leader and continuous innovator in the cutting-edge commerce distribution of electronic components and automation products worldwide, providing more than 15.3 million components from over 2,900 quality name-brand manufacturers. Read what he has to say.

The Importance of Temperature Monitoring and Accuracy During Cold and Flu Season | by Dr. Audrey Sue Cruz
What consumers and health professionals may not realize is the critical importance of utilizing proper temperature monitoring tools, to stay informed about their health, and also to evaluate the severity of illness, helping to determine if medical attention is needed. Learn more.
FDA Medical Device Updates
Market Reports
Huxley Medical’s SANSA Home Sleep Apnea Test Gets FDA Nod
In a clinical trial involving 340 patients across seven institutions, SANSA obtained its initial FDA clearance in 2024, demonstrating its accuracy in comparison to the gold-standard polysomnography. This innovative test incorporates nine distinct physiological channels, such as oximetry, respiratory effort, sleep/wake staging, and a reference electrocardiogram (ECG), into a single patch worn on the chest. This integration allows for a comprehensive understanding of cardiopulmonary health and provides valuable insights.
RapidAI Secures FDA 510(k) Clearance for Groundbreaking AI 3D Head and Neck CTA Imaging: Lumina 3D™
RapidAI continues to pioneer AI advances by revolutionizing 3D imaging with no-click, AI-powered reconstruction.
Executives

Olga Uspenskaya-Cadoz Appointed Chief Medical Officer of VectorY Therapeutics
Dr. Uspenskaya-Cadoz brings two decades of experience in neurology, gene therapy, and clinical development, enhancing VectorY’s leadership in developing transformative treatments for neurodegenerative conditions.
Operation Warp Speed Leader Tapped as Panther Life Sciences’ New R&D Chief | The Appointment of Dr Matthew Hepburn to Expand Panther’s Leadership For Next Generation Immune-Mediated Therapies
In this role, Dr Martin Hepburn will drive Panther’s strategic commitment to commercialize its patented MAP technology, with a primary focus on reducing time-to-market and delivering the first commercial product to customers within the next two years.
Michael Andersen Joins Vektor Medical as Vice President, Quality Assurance and Regulatory Affairs
Michael Andersen brings more than 20 years of commercial leadership experience in electrophysiology and medtech, with a strong track record of market development, sales execution, and strategic growth.
Advancements In Imaging

Vieworks Showcases X-ray Imaging Excellence: RSNA 2023 Spotligh
Vieworks will present its comprehensive range of DR flat panel detectors (FPDs) catering to general radiography, dynamic applications, and mammography. The focal point remains on the latest VIVIX-S F series, accompanied by the revolutionary VIVIX-S 1751S—a long-format detector tailored for imaging larger body parts and long bones.
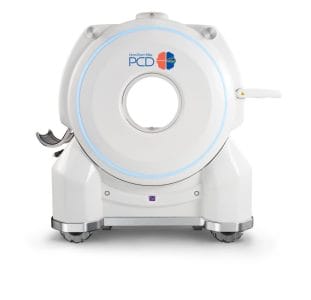
Samsung Ushers in a New Era of Diagnostic Solutions at RSNA 2023
The clinical launch of the OmniTom Elite with PCD, the first US FDA 510(k) cleared single-source photon counting computed tomography scanner with a single detector on a mobile system, will be featured at this year’s meeting. Photon counting is a next-generation CT technology that sorts the different energies of X-rays after they have passed through the scan field, capturing the X-ray photons directly at the detector and converting the energy to electrical signals.
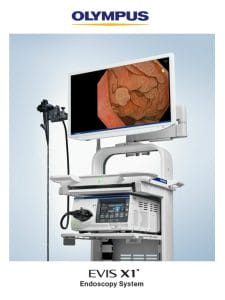
Olympus Announces Market Launch of EVIS X1 Endoscopy System
The EVIS X1 endoscopy system received clearance earlier this year along with two compatible endoscopes: the GIF-1100 gastrointestinal videoscope indicated for use within the upper digestive tract and the CF-HQ1100DL/I colonovideoscope indicated for use within the lower digestive tract.
Hospitals In the News
Non-Profit News
Healthcare: Understanding the Business Side of the Industry and Its Implications

5 Pieces of Medical Equipment Every Assisted Living Home Should Have
One of the biggest benefits of opting to have a loved one cared for in an assisted living facility is that you’re ensuring they receive better medical support than you’d be able to provide in the event of an emergency.

Fortified and Efficient: Maximizing Security Measures
Digital systems contain sensitive information, ranging from confidential business plans to private customer details, making security an essential requirement. However, excessive protections that impede legitimate usage also impair productivity, highlighting the need to balance robust barriers against unauthorized access with convenient workflows for sanctioned personnel. The most effective security measures fuse fortification against external and internal threats with efficiency for operational objectives and authorized workflows.

Incentives for Practitioners with Medicare and Medicaid Patients
Value-based programs provide financial incentives to healthcare practitioners providing care to Medicare and Medicaid patients. What Is a Value-Based Care Model? Value-based care models encourage

How to Keep Your Medical Staff Safe in Your Practice
While providing care to your patients, your practice shouldn’t neglect the need to keep your medical staff safe as they perform their duties.