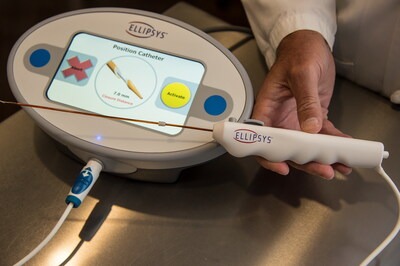
Ellipsys Medical, Inc. today announced that the Ellipsys® Vascular Access System will once again be commercially available, with initial availability anticipated in mid-2026. The announcement marks the planned reintroduction of the minimally invasive technology to clinicians and patients following a period of limited market availability. The Ellipsys System, previously available...
Medical Device News Magazine is committed to delivering high‑quality, in‑depth medical device news and biotechnology updates that include expert bylines. Our coverage offers a comprehensive view of the trends, innovations, and breakthroughs shaping the future of medical technology.
We also strive to provide a distinctive perspective on the rapidly evolving healthcare landscape. By staying connected with our reporting, you gain access to exclusive insights on industry developments, emerging technologies, and global regulatory changes that influence the medical device sector. At the same time, you join a vibrant community of professionals, innovators, and thought leaders dedicated to advancing healthcare through cutting‑edge solutions.
We invite you to explore our wide range of content—from the latest product announcements to detailed analyses of industry trends. And if our mission aligns with your goals, we offer opportunities for advertising and collaboration, helping you connect with a highly engaged audience of medical technology professionals.
Definition medical devices.
Medical Device Industry News | Latest Developments, Breakthroughs & Future Outlook
Latest News and Developments
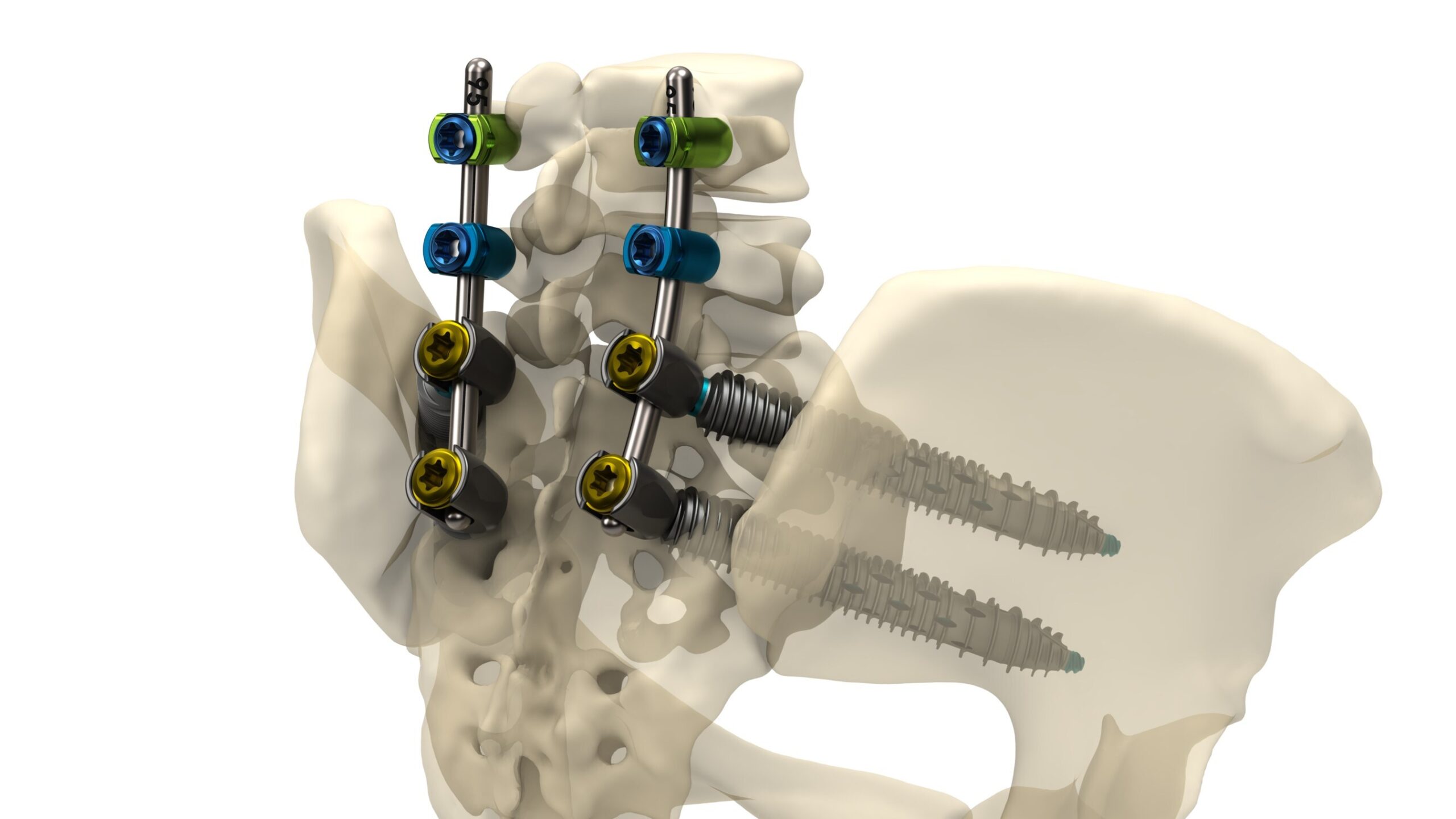
Genesys Spine Announces Launch of the SIros-X™ System for Sacroiliac Joint Fusion and Pelvic Fixation of Pedicle Screw Constructs
Inaugural case performed by spine surgeon Dr. Scott L. Parker highlights the system's strength, versatility, and precision for supporting multi-level spinal constructs
Cartessa Aesthetics and Classys Introduce QUADESSY, a Faster, Safer and More Versatile Microneedling Radiofrequency Platform
Quadessy delivers multi-layer skin remodeling through a unique tip and dual-depth design
Clinical Trials
Complement Therapeutics Announces First Patient Dosed with CTx001 in the Phase I/II Opti-GAIN Study for Geographic Atrophy Secondary to AMD
First patient dosed in Opti-GAIN, a first-in-human Phase I/II clinical trial of CTx001 in Geographic Atrophy secondary to AMD
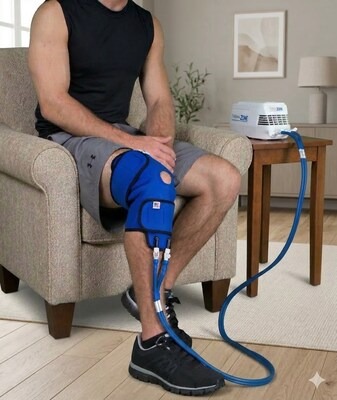
Innovative Medical Equipment, LLC Highlights Published VA Clinical Study on Thermal Therapy and Pain Treatment Utilization
The study, published in The March 2026 edition of Federal Practitioner, reported an association between thermal therapy use and reduced opioid and benzodiazepine utilization, as well as lower healthcare utilization in the patient population.
Results from PARADIGM Phase 2b Trial of PrimeC in ALS Demonstrating Meaningful Clinical Outcomes and Biological Activity Published in JAMA Neurology
The Journal of the American Medical Association (JAMA Neurology) publication highlights consistent clinical and biomarker findings from NeuroSense PARADIGM Phase 2b trial in ALS, including slower functional decline, reduced risk of ALS-related complications, and modulation of disease-relevant biomarkers
Biotechnology News
iXCells Biotechnologies Joins International Collaboration to Evaluate Chemotherapeutic Toxicity in Human Organoids
Joint initiative with Rosebud Biosciences, Kantify, and funded by Incite will identify and quantify off-target toxicities in chemotherapy patients
Primerdesign Partners with Origin Sciences to Create Novel Rectal Mucosal Extraction Workflow
Robust DNA extraction supports earlier detection and diagnosis of colorectal cancer and other gastrointestinal diseases
AnalytiChem Launches New Legionella Testing Selective Agar Plate
Redipor® Legionella agar plate range - specifically designed to support the detection and enumeration of Legionella in water samples
Mergers & Acquisitions
Funding
Apheros Secures $1.85M
Apheros notes the pre-seed funding round, led by venture capital firm Founderful, will accelerate development and deployment of the companys’ revolutionary metal foam-based cooling solutions.
MolecuLight Secures $11.7 Million in Financing to Accelerate Global Expansion
“MolecuLight’s innovative technology has the potential to significantly improve patient outcomes and offers compelling benefits for all healthcare system stakeholders,” said Guillermo Freire, Senior Vice-President, Mid-Market Group, EDC. “We are excited to invest in MolecuLight as they accelerate commercialization. EDC is focused on supporting emerging Canadian innovators in critical sectors essential for the future. This investment reflects our belief in innovation and progress, underscoring mission is to support Canadian companies in their journey to become international leaders in their sectors.”
Industry Expert Bylines
It’s Time to Move Beyond Mere Patient Navigation | By Craig Parker, JD, CPA and CEO, Guideway Care
Parker writes, “Is your patient navigation program delivering the outcomes you desire? Consider the following questions as you evaluate your current program. Read on.

Transforming Hammertoe Procedures: Forma Medical’s OptimalHT Revolutionizes MIS Hammertoe Arthrodes | By Andrew C. Davison
As the President and CEO of Forma Medical, I am excited to share how our groundbreaking approach, known as OptimalHT, is set to revolutionize hammertoe treatment. Our mission at Forma Medical has always been clear: to enhance the lives of patients by providing innovative surgical solutions. Today, I want to take you on a journey through the challenges of hammertoe deformities, the birth of OptimalHT, its key benefits, and our commitment to advancing surgery.

A Platform Approach: Expanding a Molecular Diagnostic Device Beyond Human Applications | By Shaun Holt, CEO, Alveo Technologies
Holt writes, “Avian flu is a global crisis, and not just for the poultry industry. There are serious potential implications for human health as well. But, let’s start with agriculture. Some strains of avian flu can be highly pathogenic. Once a single bird shows symptoms the clock starts ticking — it’s not unusual for an entire flock of tens of thousands of birds to die within two to three days.”
FDA Medical Device Updates
Market Reports
Executives
Advancements In Imaging
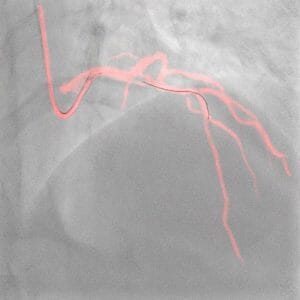
Philips’ Image-guided Navigation Increases Safety During Coronary Interventions and Reduces the Use of Contrast Media by an Average of 28.8%
The results were presented today at the European Association of Percutaneous Cardiovascular Interventions (EuroPCR) 2023 course and will also be presented later this week at the Society for Cardiovascular Angiography & Interventions (SCAI) 2023 scientific sessions.
Olympus Set to Demonstrate Newest Endoscopy System at DDW
Olympus advises the EVIS X1 endoscopy system recently received FDA 510(k) clearance along with two compatible gastrointestinal endoscopes: the GIF-1100 gastrointestinal videoscope indicated for use within the upper digestive tract and the CF-HQ1100DL/I colonovideoscope indicated for use within the lower digestive tract, which will also be displayed.
Varex Launches XRD 3131N X-Ray Detector for High Throughput Electric Vehicle Battery Inspection
XRD 3131N X-Ray Detector enables a rapid and more thorough inspection process.
Hospitals In the News
Non-Profit News
Healthcare: Understanding the Business Side of the Industry and Its Implications

Maximizing Healthcare IT Consulting for Streamlined Operations
Some outside the information technology community fear that the increasing use of computers in healthcare will unnecessarily complicate patient care. However, it’s the goal of

The Secrets of Dental Office Upgrades: 6 Tips for a Thriving Practice
The dental industry has evolved significantly over the years, with advancements in technology, patient expectations, and the competitive landscape pushing dental practitioners to constantly improve

Impact of Technology on Medical Advancements
When technology and medicine come together, it means that in the future, diseases will be found early and treated more effectively. Care for patients is getting better and better because of this vital relationship.

What Are the Top Skills Every Future Doctor Needs to Perfect?
From the delicate art of suturing to mastering advanced communication, in this article we’ll explore the multifaceted world of modern medicine.