These awards are given to the top-performing Medline distribution centers in the United States across its four tiers of size, as determined by the sales volume distributed out of each facility
Medical Device News Magazine is committed to delivering high‑quality, in‑depth medical device news and biotechnology updates that include expert bylines. Our coverage offers a comprehensive view of the trends, innovations, and breakthroughs shaping the future of medical technology.
We also strive to provide a distinctive perspective on the rapidly evolving healthcare landscape. By staying connected with our reporting, you gain access to exclusive insights on industry developments, emerging technologies, and global regulatory changes that influence the medical device sector. At the same time, you join a vibrant community of professionals, innovators, and thought leaders dedicated to advancing healthcare through cutting‑edge solutions.
We invite you to explore our wide range of content—from the latest product announcements to detailed analyses of industry trends. And if our mission aligns with your goals, we offer opportunities for advertising and collaboration, helping you connect with a highly engaged audience of medical technology professionals.
Definition medical devices.
Medical Device Industry News | Latest Developments, Breakthroughs & Future Outlook
Latest News and Developments
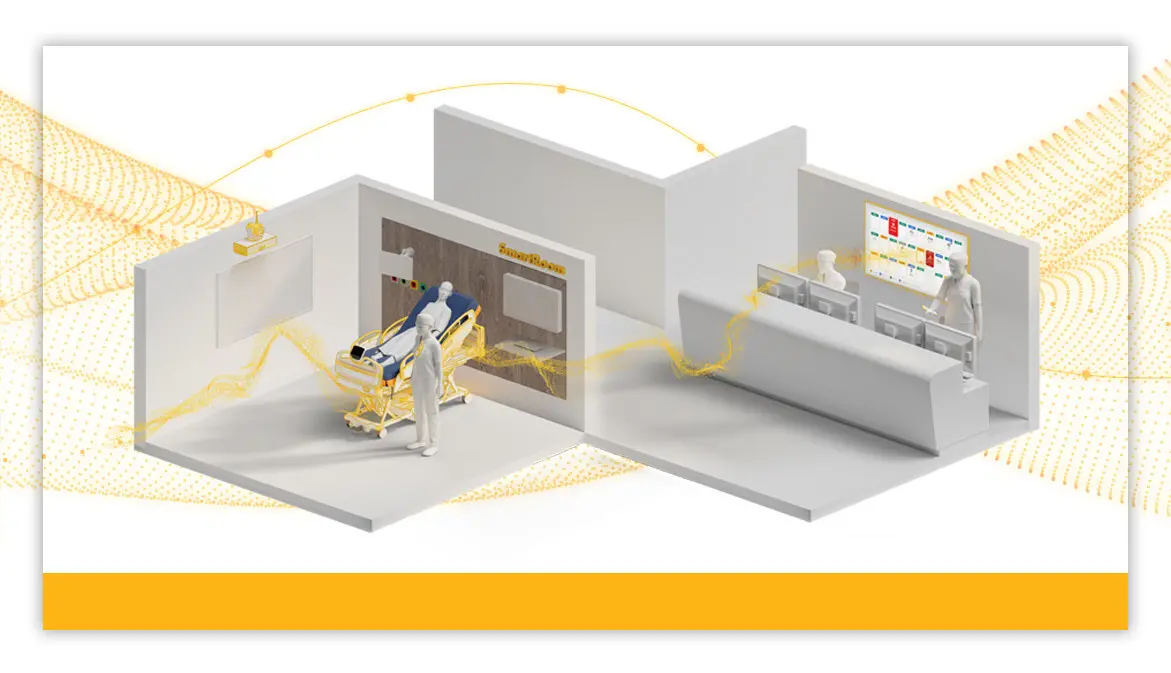
Stryker launches SmartHospital Platform
"We are dedicated to partnering with our customers on their digital journeys to help elevate care delivery," said Scott Sagehorn, VP/GM of Smart Care at Stryker. "The SmartHospital Platform is designed to evolve alongside health systems so teams can work more efficiently and stay focused on patient-centered care."
1st Patient Treated with ResQFoam for Life-Threatening Abdominal Bleeding | Reports Arsenal Medical
FDA Breakthrough-designated device under evaluation for treatment of life-threatening abdominal bleeding at leading U.S. trauma centers shows promising results in first patient
Clinilabs Establishes EU Headquarters in Basel, Strengthening European Clinical Operations Under New Regional Leadership
The company also announced the appointment of Dr. Anne-Marie Nagy as executive vice president and head of Clinilabs Europe.
CharmHealth Advances Its AI Strategy With MCP Server
By embedding AI as core infrastructure rather than an add-on, CharmHealth is advancing a more practical, governed approach to AI adoption with the EHR
Primera Selects Cenevo’s Labguru to Transition from Paper to Digitalized Workflow
“Beyond our specialized CRO solution set, our client-facing team has extensive real-world CRO experience,” said Keith Hale, CEO of Cenevo. “Our tailored solution and industry experience will help guide Primera through the steps they need to fully embrace automation and digitalization in their highly specialized environment.”
Clinical Trials
MBX Biosciences Announces Successful End-of-Phase 2 FDA Meeting and Provides Phase 3 Development Plan for Once-Weekly Canvuparatide for Hypoparathyroidism
Following End-of-Phase 2 meeting, MBX remains on track to initiate Phase 3 in Q3 2026
Medicus Pharma Provides Interpretation of Positive Phase 2 SkinJect™ Dataset
The Company Reported 73% Clinical Clearance in the 200-µg cohort at Day 57
Medicus Pharma reports positive Phase 2 SKNJCT-003 Topline Data observing 73% Clinical Clearance and 40% Histological Clearance at day 57
The company is expected to finalize the Clinical Study Report (CSR) in Q2 2026 to support planned end of Phase 2 (EOP2) meeting with the FDA
Biotechnology News
Miniature laser technology could bring lab testing into your home
By studying how various biomolecules interact with each other – for example antibodies in the immune system and xenobiotic antigens – researchers can gain valuable insights leading to new medicines and vaccines or assess whether a sample contains signs of infection
Evinova Announces Strategic Collaborations with Astellas, AstraZeneca and Bristol Myers Squibb Advancing Its AI-Native Platform to Accelerate Global Clinical Development
Each partner company has opted in to share operational data with Evinova, enabling the platform to provide benchmarks and smarter recommendations intended to further accelerate clinical trials and improve patient outcomes
Cenevo Advances Toward Agentic Labs with Two New AI Agents
The new agents are the AI Protocol Conversion agent and the AI Automation agent.