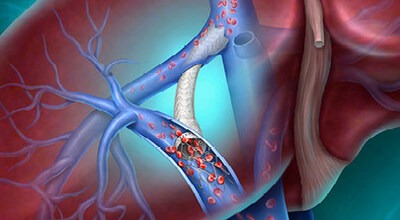
CE Marking and ARCH clinical trial data position the Liverty™ TIPS Stent Graft to expand treatment options for patients with advanced liver disease
Medical Device News Magazine is committed to delivering high‑quality, in‑depth medical device news and biotechnology updates that include expert bylines. Our coverage offers a comprehensive view of the trends, innovations, and breakthroughs shaping the future of medical technology.
We also strive to provide a distinctive perspective on the rapidly evolving healthcare landscape. By staying connected with our reporting, you gain access to exclusive insights on industry developments, emerging technologies, and global regulatory changes that influence the medical device sector. At the same time, you join a vibrant community of professionals, innovators, and thought leaders dedicated to advancing healthcare through cutting‑edge solutions.
We invite you to explore our wide range of content—from the latest product announcements to detailed analyses of industry trends. And if our mission aligns with your goals, we offer opportunities for advertising and collaboration, helping you connect with a highly engaged audience of medical technology professionals.
Definition medical devices.
Medical Device Industry News | Latest Developments, Breakthroughs & Future Outlook
Latest News and Developments
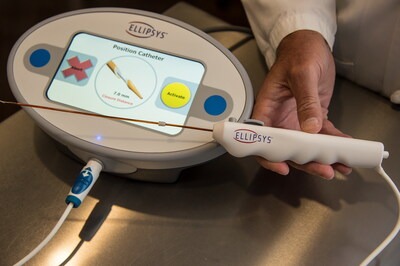
Ellipsys Vascular Access System to Return to U.S. Market in 2026
Ellipsys Medical, Inc. today announced that the Ellipsys® Vascular Access System will once again be commercially available, with initial availability anticipated in mid-2026. The announcement marks the planned reintroduction of the minimally invasive technology to clinicians and patients following a period of limited market availability. The Ellipsys System, previously available...
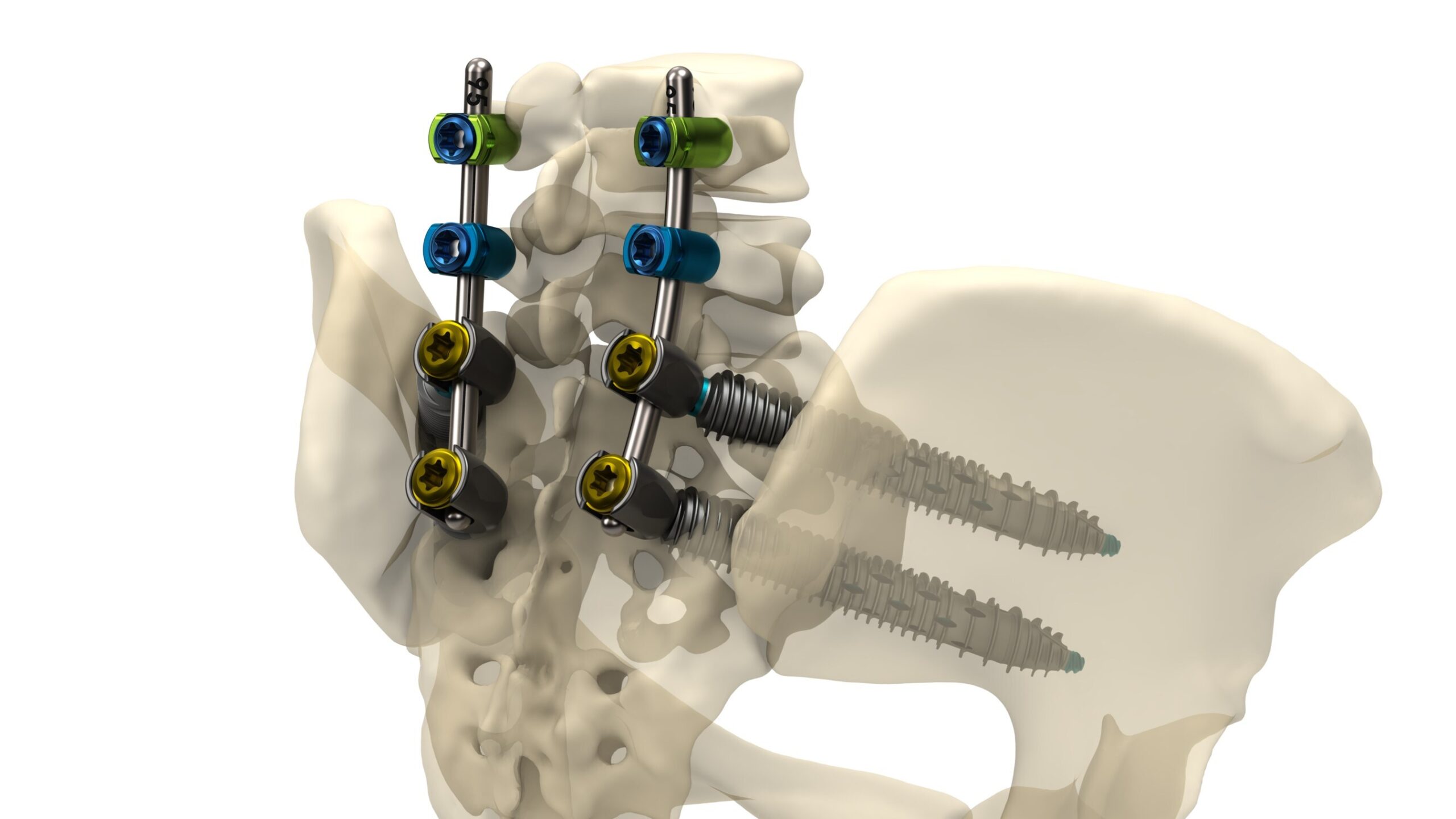
Genesys Spine Announces Launch of the SIros-X™ System for Sacroiliac Joint Fusion and Pelvic Fixation of Pedicle Screw Constructs
Inaugural case performed by spine surgeon Dr. Scott L. Parker highlights the system's strength, versatility, and precision for supporting multi-level spinal constructs
Clinical Trials
Medicus Pharma Submits Optimized Phase 2 Study Protocol to U.S. FDA for Teverelix in Acute Urinary Retention
Mechanism-Driven Study Design Focused on Capital Efficiency and Accelerated Development for Near-Term Value Creation, Addressing a $2 Billion Potential Target Market
Complement Therapeutics Announces First Patient Dosed with CTx001 in the Phase I/II Opti-GAIN Study for Geographic Atrophy Secondary to AMD
First patient dosed in Opti-GAIN, a first-in-human Phase I/II clinical trial of CTx001 in Geographic Atrophy secondary to AMD
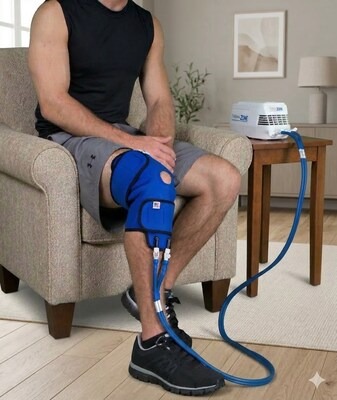
Innovative Medical Equipment, LLC Highlights Published VA Clinical Study on Thermal Therapy and Pain Treatment Utilization
The study, published in The March 2026 edition of Federal Practitioner, reported an association between thermal therapy use and reduced opioid and benzodiazepine utilization, as well as lower healthcare utilization in the patient population.
Biotechnology News
Variational AI Releases Enki 4: Major Update to Foundation Model for Small-Molecule Drug Discovery
Variational AI introduces Enki™ 4: an improved algorithm and architecture designed to scale, expanding the pre-trained target coverage from 592 to 760 and now applicable to proximity-based therapeutics and novel payloads for antibody drug conjugates
Transgene and NEC Bio Sign License Agreement to Prepare the Next Steps of the Development of TG4050 in Head and Neck Cancer
Akira Kitamura, GM, AI Drug Development Division of NEC Corporation and CEO of NEC Bio, added, "This agreement is an important milestone in our partnership with Transgene and reflects NEC’s long-term commitment to the development of TG4050
iXCells Biotechnologies Joins International Collaboration to Evaluate Chemotherapeutic Toxicity in Human Organoids
Joint initiative with Rosebud Biosciences, Kantify, and funded by Incite will identify and quantify off-target toxicities in chemotherapy patients
Mergers & Acquisitions
Funding
U.S. Department of Defense Awards Theradaptive $4 Million Contract for Its OsteoAdapt Regenerative Therapeutic Program
The funds from the DOD contract will enable Theradaptive to continue its work to meet regulatory requirements and scale up Good Manufacturing Practices-compliant manufacturing of the OsteoAdapt product in preparation for clinical studies.
Macrolux Completes Nearly CNY100 million Series A Round: Funding Led by Legend Capital
Legend Capital has long been paying attention to the investment in the field of medical devices and diagnostics with a focus on three major themes: first, major diseases, especially the unmet clinical needs of chronic diseases; second, the first and uniqueness in technology, which enables a company to become or have the potential to become a leading player in subdivided industries; the third is the “device +” investment opportunities brought by the integration of medical devices and interdisciplinary technologies such as AI, IoT and Big Data.
Industry Expert Bylines

Back to the Future: Exploring the Past, Reviewing the Present, and Planning for the Future of Infection Prevention
By: Caitlin Stowe, MPH, CPH, CIC, CPHQ, VA-BC, Vice President of Clinical Affairs and Medical Liaison, ActivePure Medical.

Postgame Video Review Elite Athletes Experience is Now Possible for Surgeons
By George Murgatroyd, Vice President and General Manager, Digital Surgery, Surgical Robotics, Medtronic writes, “AI-powered video review technology helps athletes at nearly every competitive level make the subtle changes that can be the difference between winning and losing a game. It’s surprising that surgeons, who often operate using video camera systems, don’t benefit from the same opportunity to review their cases.” Read on to learn more!

Patient-First Model Improves Care, Maximizes Therapeutic Opportunities for Patients with Rare Diseases – By Michelle Hefley, Co-Founder, Managing Partner and Chairman, Optime Care
Michelle Hefley writes, “During Patient Experience Week, it’s important for more people to understand the challenges that rare disease patients face and how a patient-first strategy to specialty drug management offers a more compassionate way to provide better services and support.” Read on to learn more.