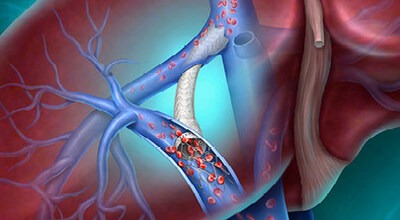
CE Marking and ARCH clinical trial data position the Liverty™ TIPS Stent Graft to expand treatment options for patients with advanced liver disease
Medical Device News Magazine is committed to delivering high‑quality, in‑depth medical device news and biotechnology updates that include expert bylines. Our coverage offers a comprehensive view of the trends, innovations, and breakthroughs shaping the future of medical technology.
We also strive to provide a distinctive perspective on the rapidly evolving healthcare landscape. By staying connected with our reporting, you gain access to exclusive insights on industry developments, emerging technologies, and global regulatory changes that influence the medical device sector. At the same time, you join a vibrant community of professionals, innovators, and thought leaders dedicated to advancing healthcare through cutting‑edge solutions.
We invite you to explore our wide range of content—from the latest product announcements to detailed analyses of industry trends. And if our mission aligns with your goals, we offer opportunities for advertising and collaboration, helping you connect with a highly engaged audience of medical technology professionals.
Definition medical devices.
Medical Device Industry News | Latest Developments, Breakthroughs & Future Outlook
Latest News and Developments
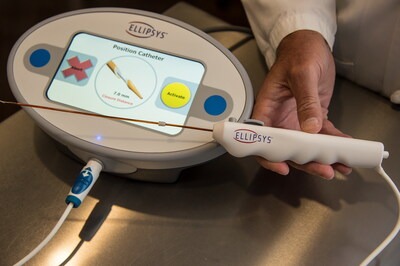
Ellipsys Vascular Access System to Return to U.S. Market in 2026
Ellipsys Medical, Inc. today announced that the Ellipsys® Vascular Access System will once again be commercially available, with initial availability anticipated in mid-2026. The announcement marks the planned reintroduction of the minimally invasive technology to clinicians and patients following a period of limited market availability. The Ellipsys System, previously available...
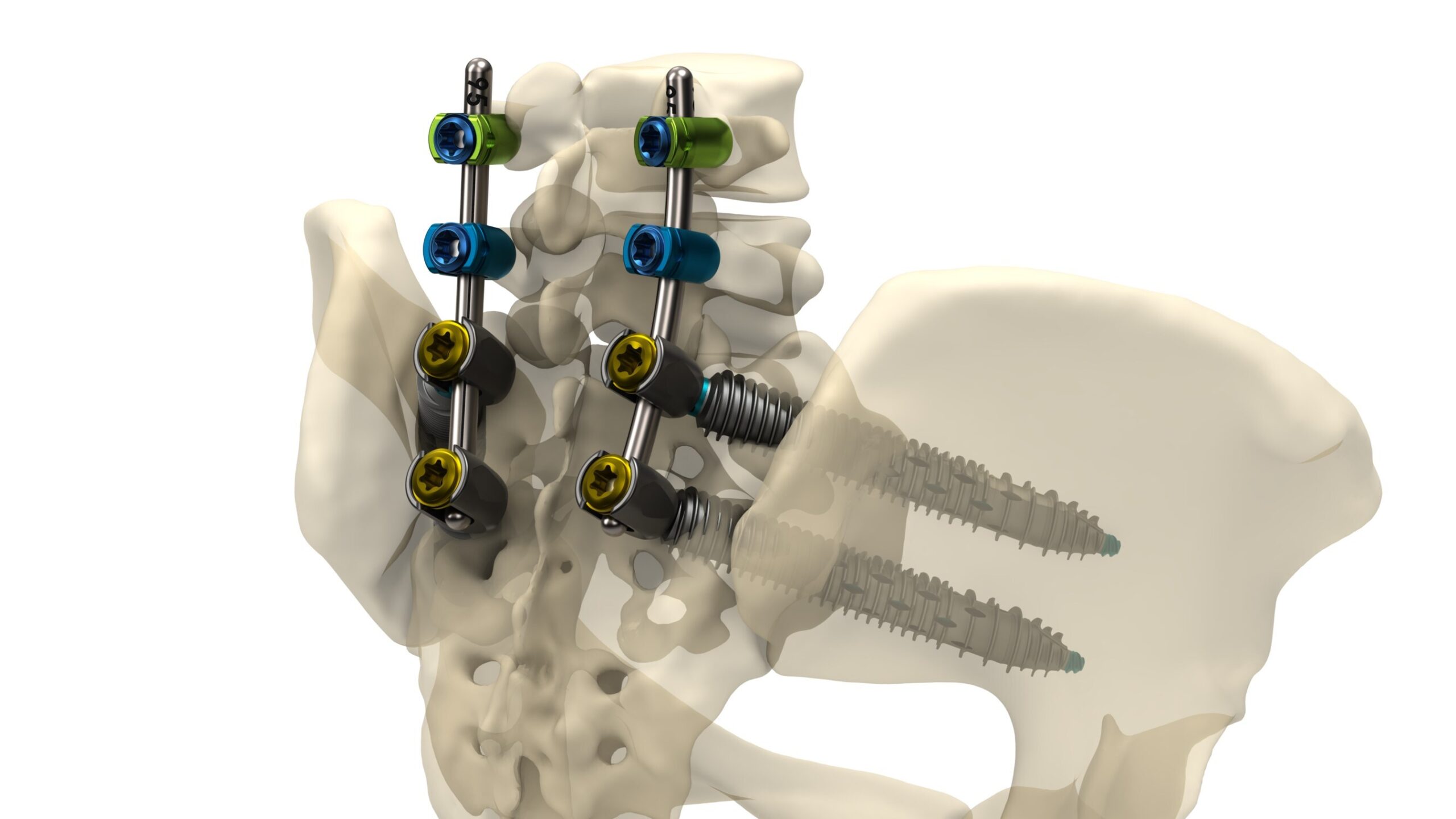
Genesys Spine Announces Launch of the SIros-X™ System for Sacroiliac Joint Fusion and Pelvic Fixation of Pedicle Screw Constructs
Inaugural case performed by spine surgeon Dr. Scott L. Parker highlights the system's strength, versatility, and precision for supporting multi-level spinal constructs
Clinical Trials
First Implant of KingstronBio’s ProStyle M® Transcatheter Mitral Valve System Successfully Completed in National Multicenter Confirmatory Study
On March 12, 2026, the first implant of the ProStyle M® Transcatheter Mitral Valve System (ProStyle M®) was successfully completed under pure ultrasound guidance as part of its confirmatory clinical study. The device is independently developed by KingstronBio Technology (Changshu) Co., Ltd. (KingstronBio). The procedure was performed by Professor Wang Chunsheng and Professor...
Supira Medical Announces FDA Approval for SUPPORT II Pivotal Trial, Advances in Cardiogenic Shock
Company Appoints D. Keith Grossman to Board of Directors
Medicus Pharma Submits Optimized Phase 2 Study Protocol to U.S. FDA for Teverelix in Acute Urinary Retention
Mechanism-Driven Study Design Focused on Capital Efficiency and Accelerated Development for Near-Term Value Creation, Addressing a $2 Billion Potential Target Market
Biotechnology News
Variational AI Releases Enki 4: Major Update to Foundation Model for Small-Molecule Drug Discovery
Variational AI introduces Enki™ 4: an improved algorithm and architecture designed to scale, expanding the pre-trained target coverage from 592 to 760 and now applicable to proximity-based therapeutics and novel payloads for antibody drug conjugates
Transgene and NEC Bio Sign License Agreement to Prepare the Next Steps of the Development of TG4050 in Head and Neck Cancer
Akira Kitamura, GM, AI Drug Development Division of NEC Corporation and CEO of NEC Bio, added, "This agreement is an important milestone in our partnership with Transgene and reflects NEC’s long-term commitment to the development of TG4050
iXCells Biotechnologies Joins International Collaboration to Evaluate Chemotherapeutic Toxicity in Human Organoids
Joint initiative with Rosebud Biosciences, Kantify, and funded by Incite will identify and quantify off-target toxicities in chemotherapy patients
Mergers & Acquisitions
Funding

Hexarad Completes Series A Funding – Raises Additional £2.2 Million
The additional funding raised through this round will allow Hexarad to continue to grow its radiology platform which focuses on allocative efficiency to improve scan reporting times.
German Federal Ministry of Education and Research Awards Grants to Gund a Clinical Trial for LISA Laser Innovated “Novel Treatment of Urolithiasis”
Urolithiasis is a common disease affecting about 12% of the global population. Consequently, urolithiasis is a widespread disease with significant socioeconomic impact and is relevant to healthcare in terms of society and health policy.
Industry Expert Bylines

Testing the Benefits of Module-Based Simulation Training for Arthroscopic Surgery
Samuel Larrivée, MD, MSc, FRCSC notes it’s no secret that complex arthroscopic procedures can be difficult to teach to residents and experienced doctors alike. Read on.
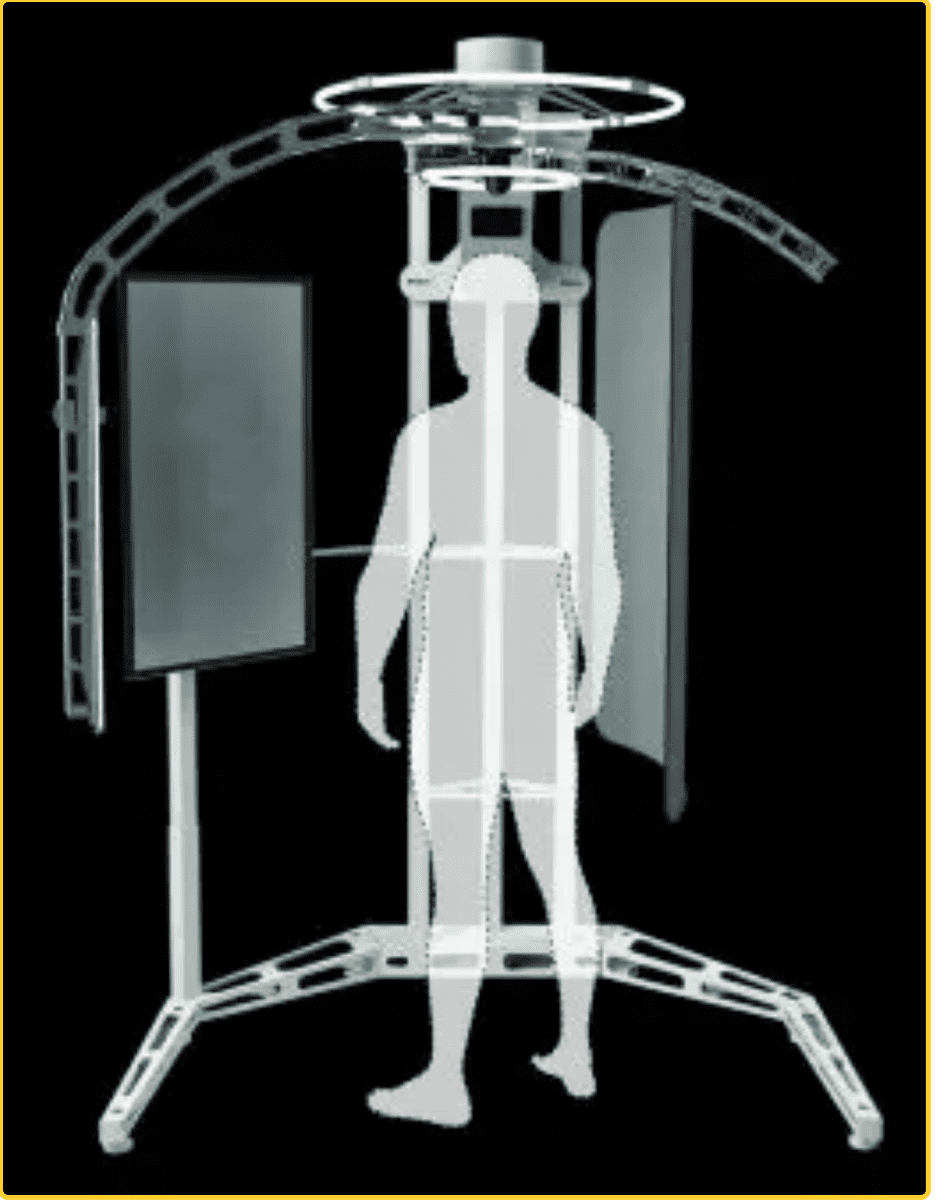
oVio’s New AI Enhanced, Fully Dimensional Imaging Technology May Be a Game Changer for Healthcare & Security Sectors
oVio’s imaging technology has been heavily used and adopted by medical practitioners in cosmetic and elective surgery specialties, allowing for patients to have 360-degree views of everything from before-and-afters to prospective post-op results. Their existing products are well-established as an invaluable resource to doctors providing cosmetic surgery, skincare, hair replacement treatment, and other services to their clients.
Racing Towards UKCA Compliance: Expert Ed Ball, Senior Associate, RQM+ – A Professionals View
The UK will continue with the UK Medical Devices Regulations 2002 (S.I. 2002/618) (UK MDR), as amended, and by the 30th of June 2023, manufacturers will need to follow the UK Conformity Assessment process and place the UKCA mark on their medical devices, as CE marked medical devices will stop being recognised in Great Britain (England, Scotland and Wales).