January 7, 2021
QT Imaging notes Freedom Ventures, is a Dutch company that will do business as QT Scan BV, is well established in the field of medical devices, with an extensive network of distributors, and a Center of Excellence for technical, marketing, and clinical training.
“As a small company, working with an established distribution network provides our global market reach while being able to maintain the level of quality in marketing, sales, technical service, and most importantly patient care, that we have established in the US. Freedom Ventures has the experience to be that partner in this important region,” says Meg Donigan, Chief Operating Officer at QT Imaging. Freedom Ventures will also assist QT Imaging in securing a CE Mark and manage the registration process in its markets.

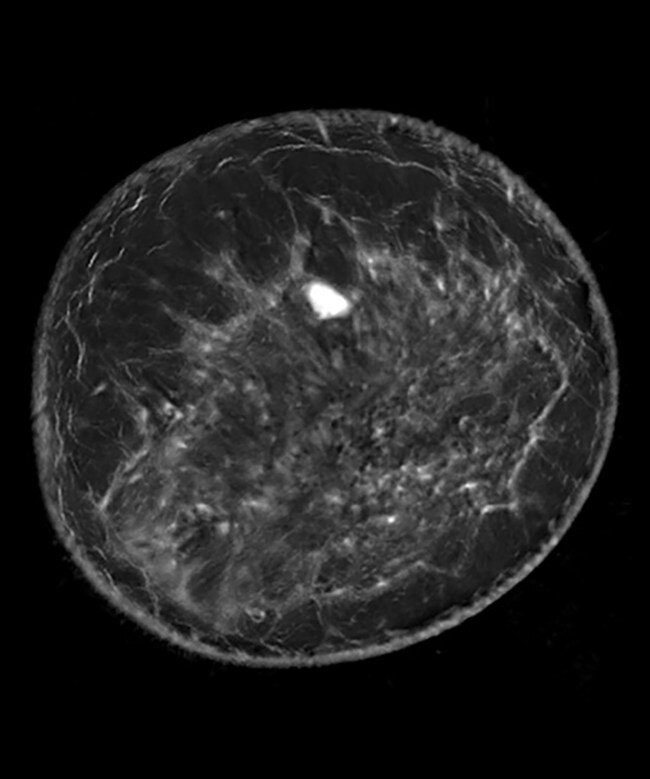
QT’s medical imaging technology generates 3D images with remarkable accuracy and precision, and the volumetric images are produced without the need of contrast injections or radiation. The QTscan® is an FDA-cleared breast imaging tool, and the company will seek CE marking to provide confidence to the healthcare community that the device meets EU safety, health, and environmental protection requirements before market launch.
Rutger Brest van Kempen, CEO of Freedom Ventures, says “We’re very excited about the opportunity the radiation-free QTscan has to fill a unique need in the European breast imaging market where women are being screened later and less frequently due to concerns about radiation. In the Middle East and North Africa, this new technology will also be embraced for its ease of adoption as it requires no costly facilities or special licensing for operators.
Hisham Hasaan, Freedom Ventures Vice-President for the Middle East and Africa says, “With QTscan we will have a huge opportunity to screen woman, who now have to travel far to hospitals where radiology equipment is available whereas our technologies can be installed virtually anywhere. We will focus on creating regional QTscan centers to bring this unique technology to the masses.”