TIGER TRIAL: The first adjustable, fully visible stent retriever shows superiority over conventional stent retrievers in a multicenter trial.
The late-breaking results were recently presented at the International Stroke Conference and published online in the journal Stroke.
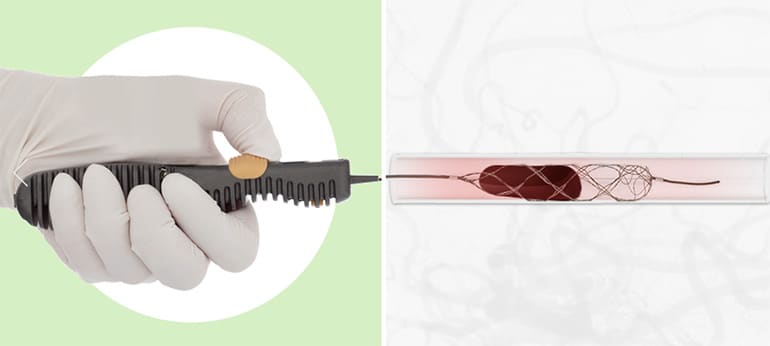
The TIGER Trial demonstrates a statistically significant 11-point improvement in the ability to restore blood flow to the brain during ischemic stroke and equivalent safety for the TIGERTRIEVER™ device compared to existing self-expanding stent retrievers.
“The trial confirmed that the TIGERTRIEVER is not only safe and effective but demonstrably improved outcomes over similar trials testing previous technology,” observed Jeffery Saver, MD, FAHA, Professor of Neurology and Director of the Comprehensive Stroke and Vascular Neurology Program at UCLA. “TIGERTRIEVER is an important advance in stroke treatment devices and a win for patients.”
Stent retriever devices (“stentrievers”) are the gold standard in treating large vessel ischemic strokes. Delivered minimally invasively through the vasculature, they expand into the clot to capture and remove the blockage as the device is retrieved. Prior to TIGERTRIEVER, the diameter and force of these devices were non-adjustable. With TIGERTRIEVER, physicians control the diameter and force as needed in response to real-time anatomical and procedural information. This ability to respond to situational feedback is known as intelligent control. The TIGER Trial concludes that these improvements provide meaningful benefit when removing the clot and treating stroke.
The TIGER Trial ended early after overwhelming effectiveness with 160 patients enrolled at 17 centers in the US and Israel. The trial demonstrates 84.6% revascularization success, compared to a 73.4% average from 6 recent pivotal stent retriever trials. This improvement translates to significantly better patient outcomes, with 58% of patients living independently after 90 days versus an average of 43% of patients from the previous device trials. Additionally, the TIGER Trial shows improved safety, with an 18.1% primary safety score versus a 20.4% historical average.
“TIGERTRIEVER’s intelligent control represents a new generation of stent retrievers as shown by the highest revascularization success rate of similar stent retriever trials,” said Walid Haddad, Chief Clinical Officer at Rapid Medical. “We applaud the work performed by our national principal investigators, Drs. Jeffery Saver of UCLA and Rishi Gupta of Wellstar Health System in Atlanta, GA, and all of our investigators and clinical site staff.”
