Quadessy delivers multi-layer skin remodeling through a unique tip and dual-depth design
Medical Device News Magazine is committed to delivering high‑quality, in‑depth medical device news and biotechnology updates that include expert bylines. Our coverage offers a comprehensive view of the trends, innovations, and breakthroughs shaping the future of medical technology.
We also strive to provide a distinctive perspective on the rapidly evolving healthcare landscape. By staying connected with our reporting, you gain access to exclusive insights on industry developments, emerging technologies, and global regulatory changes that influence the medical device sector. At the same time, you join a vibrant community of professionals, innovators, and thought leaders dedicated to advancing healthcare through cutting‑edge solutions.
We invite you to explore our wide range of content—from the latest product announcements to detailed analyses of industry trends. And if our mission aligns with your goals, we offer opportunities for advertising and collaboration, helping you connect with a highly engaged audience of medical technology professionals.
Definition medical devices.
Medical Device Industry News | Latest Developments, Breakthroughs & Future Outlook
Latest News and Developments
Medtronic and Merit Medical Systems enter into agreement to offer new, FDA-cleared ViaVerte™ system, a basivertebral nerve ablation treatment for chronic vertebrogenic lower-back pain
Medtronic's latest partnership with Merit Medical Systems introduces the groundbreaking ViaVerte™ BVNA system—an innovative solution for chronic vertebrogenic pain
Optellum AI Shown to be Highly Cost-Effective in First US Lifetime Payer Study of Lung Nodule Risk Stratification
Led by researchers at the Vanderbilt University Medical Center, a new study reports that Optellum's FDA-cleared Lung Cancer Prediction (LCP) AI assisted risk stratification for indeterminate lung nodules is cost-effective from a US payer perspective. Optellum's LCP AI supports more consistent long nodule management and helps enable earlier diagnosis of...
Clinical Trials
Complement Therapeutics Announces First Patient Dosed with CTx001 in the Phase I/II Opti-GAIN Study for Geographic Atrophy Secondary to AMD
First patient dosed in Opti-GAIN, a first-in-human Phase I/II clinical trial of CTx001 in Geographic Atrophy secondary to AMD
Innovative Medical Equipment, LLC Highlights Published VA Clinical Study on Thermal Therapy and Pain Treatment Utilization
The study, published in The March 2026 edition of Federal Practitioner, reported an association between thermal therapy use and reduced opioid and benzodiazepine utilization, as well as lower healthcare utilization in the patient population.
Results from PARADIGM Phase 2b Trial of PrimeC in ALS Demonstrating Meaningful Clinical Outcomes and Biological Activity Published in JAMA Neurology
The Journal of the American Medical Association (JAMA Neurology) publication highlights consistent clinical and biomarker findings from NeuroSense PARADIGM Phase 2b trial in ALS, including slower functional decline, reduced risk of ALS-related complications, and modulation of disease-relevant biomarkers
Biotechnology News
iXCells Biotechnologies Joins International Collaboration to Evaluate Chemotherapeutic Toxicity in Human Organoids
Joint initiative with Rosebud Biosciences, Kantify, and funded by Incite will identify and quantify off-target toxicities in chemotherapy patients
Primerdesign Partners with Origin Sciences to Create Novel Rectal Mucosal Extraction Workflow
Robust DNA extraction supports earlier detection and diagnosis of colorectal cancer and other gastrointestinal diseases
AnalytiChem Launches New Legionella Testing Selective Agar Plate
Redipor® Legionella agar plate range - specifically designed to support the detection and enumeration of Legionella in water samples
Mergers & Acquisitions
Funding
Dawn Health Secures €11.5 Million to Scale Platform & Product Suite for Next-Gen Pharma Digital Health Solutions
The investment is aimed at supporting the company’s strategy to deliver its platform and product suite to global pharma companies through a SaaS model, while continuing to invest in further offerings within the Dawn Product Suite.
Oramed Pharmaceuticals Announces $36.9 Million Investment and Strategic Collaboration with Alpha Tau Medical
Oramed leverages its strategic capital markets expertise to support Alpha Tau’s breakthrough cancer therapy technology
Industry Expert Bylines

Engineering Better Healthcare: The Vital Role of Usability Testing and HFE/UE in Medical Device Development | By Oliver Eikenberg
Both HFE and UE are based on the international usability standards IEC 62366-1 and IEC 62366-2 with consideration of risk management (ISO 14971). By planning and integrating usability testing for human interaction with medical device technology throughout the device lifecycle and risk management processes, HFE/UE ensures that medical devices meet the complex needs of healthcare professionals and patients, and that user-related and use-related risks are eliminated or mitigated.

Scaling with Demand: Requirements for Success in Medical Device Manufacturing | By Laura Beckwith, Director of Product Management, Configit
Our expert writes: Historically, medical device companies have provided value by producing equipment for diagnosing and treating patients worldwide. However, contemporary challenges such as heightened pricing pressures, regulatory compliance, supply chain volatility and operational inefficiencies have significantly changed the landscape for these companies.

Optimizing Biopharma Workflows: Integrating AI to Enhance Research and Discovery | By Christian Olsen, Associate VP & Industry Principal for Biologics at Dotmatics
The integration of AI in biopharma workflows is not just a technological advancement, but a paradigm shift that promises to completely revolutionize how we research, understand, and treat diseases. Read what this expert has to say.
FDA Medical Device Updates
Market Reports
Mursla Bio Receives FDA Breakthrough Device Designation for EvoLiver Test
Reflects the potential of EvoLiver test to improve early liver cancer detection among high-risk patients
Nanox Receives FDA Clearance for General Use of New Imaging System, Nanox.ARC X
First FDA clearance for Nanox.ARC X to produce tomographic images for general use, including musculoskeletal, pulmonary, intra-abdominal and paranasal indications. The enhanced imaging system features sleek design with smaller footprint; simplified ‘plug and play’ installation process and the system design enables software upgrades and new capabilities to be added remotely following future regulatory clearances
Executives

SynerFuse® Announces Chief Technology Officer | Michael Park, M.D., Ph.D.
Dr. Park is a former principal investigator for the SynerFuse® proof-of-concept study and primary inventor of SynerFuse® technology.
Clinilabs Expands Executive Leadership Team With Key Appointments
Clinilabs, LLC, a leading full-service contract research organization (CRO) specializing in central nervous system (CNS) drug and medical technology development, today announced the appointments of

Rad AI Names Demetri Giannikopoulos as Chief Growth Officer to Accelerate AI-Driven Radiology Healthcare Growth
Mr. Demetri brings over 20 years of experience in healthcare technology, with a track record of advancing AI adoption in complex clinical settings.
Advancements In Imaging
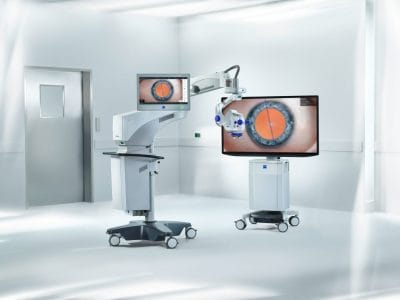
Advancing Patient Care: ZEISS Unleashes Breakthroughs in Ophthalmic Surgery at ASCRS 2024
Building on its ZEISS Medical Ecosystem, several of the new innovations enable seamless data integration and management across the cataract and refractive workflows, creating a new standard for efficient, personalized ophthalmic care.

Wireless Handheld X-ray Equipment from MinXray Used for Global Health Initiative in Papua New Guinea
MinXray MinXray, a leading manufacturer of imaging systems for medical and veterinary use, recently sent its Impact Wireless X-ray system with a group of researchers
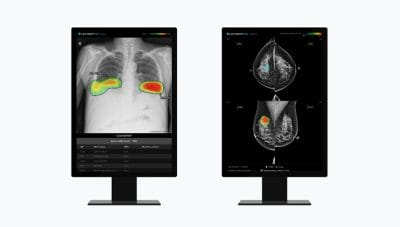
Lunit Expands Presence in Europe | To Deliver AI-powered Cancer Screening Solution to France and Portugal
New supply contract to bring Lunit INSIGHT CXR to TeleDiag in France and Lunit INSIGHT MMG to the Central region branch of the Portuguese League Against Cancer
Hospitals In the News
Non-Profit News
Healthcare: Understanding the Business Side of the Industry and Its Implications

The Benefits of Using EMR for Mental Health Practices
EMR systems enable mental health professionals to maintain and access patient records with ease, creating a more efficient workflow and better patient outcomes.

The Future of Cybersecurity: How AI and Machine Learning Are Changing the Game
As cyber threats become more sophisticated, organizations must adapt by leveraging advanced technologies to protect their data and systems. AI and ML are transforming the way cybersecurity professionals detect, respond to, and prevent cyberattacks, providing more efficient and proactive security solutions.

Protecting Surgeons’ Vision: The Critical Role of Laser Safety Glasses in Modern Operating Rooms
Surgeons face numerous risks during procedures, with eye protection being a paramount concern. Protecting surgeons’ vision is crucial, and laser safety glasses play a critical

Telemedicine: Bridging the Gap in Healthcare Access and Delivery
The healthcare industry has seen significant changes and transformations in the rapidly advancing world of technology. One of the most groundbreaking developments in recent times